Commercially Available Flavonols Are Better SARS-CoV-2 Inhibitors than Isoflavone and Flavones
- PMID: 35891437
- PMCID: PMC9324382
- DOI: 10.3390/v14071458
Commercially Available Flavonols Are Better SARS-CoV-2 Inhibitors than Isoflavone and Flavones
Abstract
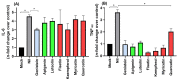
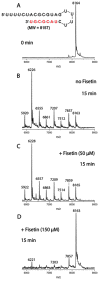
Despite the fast development of vaccines, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is still circulating and generating variants of concern (VoC) that escape the humoral immune response. In this context, the search for anti-SARS-CoV-2 compounds is still essential. A class of natural polyphenols known as flavonoids, frequently available in fruits and vegetables, is widely explored in the treatment of different diseases and used as a scaffold for the design of novel drugs. Therefore, herein we evaluate seven flavonoids divided into three subclasses, isoflavone (genistein), flavone (apigenin and luteolin) and flavonol (fisetin, kaempferol, myricetin, and quercetin), for COVID-19 treatment using cell-based assays and in silico calculations validated with experimental enzymatic data. The flavonols were better SARS-CoV-2 inhibitors than isoflavone and flavones. The increasing number of hydroxyl groups in ring B of the flavonols kaempferol, quercetin, and myricetin decreased the 50% effective concentration (EC50) value due to their impact on the orientation of the compounds inside the target. Myricetin and fisetin appear to be preferred candidates; they are both anti-inflammatory (decreasing TNF-α levels) and inhibit SARS-CoV-2 mainly by targeting the processability of the main protease (Mpro) in a non-competitive manner, with a potency comparable to the repurposed drug atazanavir. However, fisetin and myricetin might also be considered hits that are amenable to synthetic modification to improve their anti-SARS-CoV-2 profile by inhibiting not only Mpro, but also the 3'-5' exonuclease (ExoN).
Keywords: COVID-19; SARS-CoV-2; calu-3 cells; exonuclease; flavonoids; molecular docking; natural product; protease.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analysis, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures





References
-
- COVID-19 Dashboardby the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU) [(accessed on 31 May 2022)]. Available online: https://coronavirus.jhu.edu/map.html.
-
- Coronavirus (COVID-19)|Drugs. [(accessed on 15 May 2022)]; Available online: https://www.fda.gov/drugs/emergency-preparedness-drugs/coronavirus-covid....
-
- Caniels T.G., Bontjer I., van der Straten K., Poniman M., Burger J.A., Appelman B., Lavell A.H.A., Oomen M., Godeke G.-J., Valle C., et al. Emerging SARS-CoV-2 variants of concern evade humoral immune responses from infection and vaccination. Sci. Adv. 2021;7:eabj5365. doi: 10.1126/sciadv.abj5365. - DOI - PMC - PubMed
-
- Obeid M., Suffiotti M., Pellaton C., Bouchaab H., Cairoli A., Salvadé V., Stevenel C., Hottinger R., Pythoud C., Coutechier L., et al. Humoral responses against variants of concern by COVID-19 mRNA vaccines in immunocompromised patients. JAMA Oncol. 2022;8:e220446. doi: 10.1001/jamaoncol.2022.0446. - DOI - PMC - PubMed
-
- Garcia-Beltran W.F., Lam E.C., St Denis K., Nitido A.D., Garcia Z.H., Hauser B.M., Feldman J., Pavlovic M.N., Gregory D.J., Poznansky M.C., et al. Multiple SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity. Cell. 2021;184:2372–2383. doi: 10.1016/j.cell.2021.03.013. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

