A Testing Campaign Intervention Consisting of Peer-Facilitated Engagement, Point-of-Care HCV RNA Testing, and Linkage to Nursing Support to Enhance Hepatitis C Treatment Uptake among People Who Inject Drugs: The ETHOS Engage Study
- PMID: 35891535
- PMCID: PMC9316739
- DOI: 10.3390/v14071555
A Testing Campaign Intervention Consisting of Peer-Facilitated Engagement, Point-of-Care HCV RNA Testing, and Linkage to Nursing Support to Enhance Hepatitis C Treatment Uptake among People Who Inject Drugs: The ETHOS Engage Study
Abstract
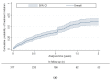
This study evaluated HCV treatment initiation among people who inject drugs (PWID) following an intervention of campaign days involving peer connection, point-of-care HCV RNA testing, and linkage to nursing support. ETHOS Engage is an observational cohort study of PWID attending 25 drug treatment clinics and needle and syringe programs in Australia (May 2018-September 2019). Point-of-care results were provided to the nurse, facilitating confirmatory testing and treatment. The study aimed to evaluate treatment uptake and factors associated with treatment at 24 months post-enrolment. There were 317 people with current HCV infection and eligible for treatment (median age 43, 65% male, 15% homeless, 69% receiving opioid agonist treatment, 70% injected in last month). Overall, 15% (47/317), 27% (85/317), 38% (120/317), and 49% (155/317) of people with current HCV infection had initiated treatment at 3-, 6-, 12-, and 24-months following testing, respectively. Homelessness (adjusted hazard ratio (aHR): 0.40; 95% confidence interval: 0.23, 0.71) and incarceration in the past 12 months (vs. never, aHR:0.46; 0.28, 0.76) were associated with decreased treatment initiation in the 24 months post-enrolment. This testing campaign intervention facilitated HCV treatment uptake among PWID. Further interventions are needed to achieve HCV elimination among people experiencing homelessness or incarceration.
Keywords: Hepatitis C virus elimination; Hepatitis C virus infection; Hepatitis C virus treatment; direct-acting antiviral era; people who inject drugs.
Conflict of interest statement
J.G. reports grants from Camurus, grants from Cepheid, grants from Hologic, grants from Indivior, grants from Merck, during the conduct of the study; grants and personal fees from Abbvie, grants and personal fees from Gilead Sciences, grants and personal fees from Merck, grants and personal fees from Cepheid, outside the submitted work. G.J.D. reports grants from Gilead, Abbvie, and Merck. L.D. has received investigator-initiated untied educational grants for studies of opioid medications in Australia from Indivior, Mundipharma and Seqirus. All remaining authors have no potential conflicts to declare. Cepheid and Merck, Sharp, and Dohme Corporation were not involved in the study design, methodology, and writing of this manuscript. The opinions expressed in the paper are those of the authors and do not necessarily represent those of Cepheid or Merck Sharp, and Dohme Corporation. The views expressed in this manuscript do not necessarily represent the position of the Australian Government.
Figures

References
-
- World Health Organization . Global Health Sector Strategy on Viral Hepatitis 2016-2021: Towards Ending Viral Hepatitis. World Health Organization; Geneva, Switzerland: 2016.
-
- Grebely J., Larney S., Peacock A., Colledge S., Leung J., Hickman M., Vickerman P., Blach S., Cunningham E.B., Dumchev K., et al. Global, regional, and country-level estimates of hepatitis C infection among people who have recently injected drugs. Addiction. 2019;114:150–166. doi: 10.1111/add.14393. - DOI - PMC - PubMed
-
- UNAIDS-Joint United Nations Programme on HIV/AIDS . Health, Rights and Drugs-Harm Reduction, Decriminalization and Zero Discrimination for People Who Use Drugs. UNAIDS; Geneva, Switzerland: 2019.
-
- Hopwood M., Treloar C., Bryant J. Hepatitis C and injecting-related discrimination in New South Wales, Australia. Drugs Educ. Prev. Policy. 2006;13:61–75. doi: 10.1080/09687630500481150. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

