Neuroprotective and Anti-Inflammatory Effects of Linoleic Acid in Models of Parkinson's Disease: The Implication of Lipid Droplets and Lipophagy
- PMID: 35892594
- PMCID: PMC9331796
- DOI: 10.3390/cells11152297
Neuroprotective and Anti-Inflammatory Effects of Linoleic Acid in Models of Parkinson's Disease: The Implication of Lipid Droplets and Lipophagy
Abstract
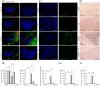
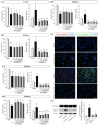
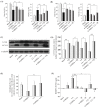
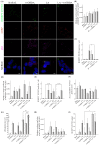
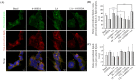
Parkinson's disease (PD) is the second most prevalent neurodegenerative disease after Alzheimer's disease. The principal pathological feature of PD is the progressive loss of dopaminergic neurons in the ventral midbrain. This pathology involves several cellular alterations: oxidative stress, mitochondrial dysfunction, loss of proteostasis, and autophagy impairment. Moreover, in recent years, lipid metabolism alterations have become relevant in PD pathogeny. The modification of lipid metabolism has become a possible way to treat the disease. Because of this, we analyzed the effect and possible mechanism of action of linoleic acid (LA) on an SH-SY5Y PD cell line model and a PD mouse model, both induced by 6-hydroxydopamine (6-OHDA) treatment. The results show that LA acts as a potent neuroprotective and anti-inflammatory agent in these PD models. We also observed that LA stimulates the biogenesis of lipid droplets and improves the autophagy/lipophagy flux, which resulted in an antioxidant effect in the in vitro PD model. In summary, we confirmed the neuroprotective effect of LA in vitro and in vivo against PD. We also obtained some clues about the novel neuroprotective mechanism of LA against PD through the regulation of lipid droplet dynamics.
Keywords: autophagy; lipid droplets; lipophagy; neurodegeneration; oxidative stress.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
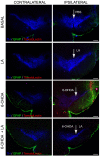
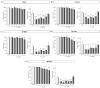
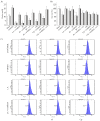
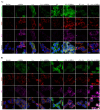


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

