Photocatalytic Degradation of Some Typical Antibiotics: Recent Advances and Future Outlooks
- PMID: 35897716
- PMCID: PMC9331861
- DOI: 10.3390/ijms23158130
Photocatalytic Degradation of Some Typical Antibiotics: Recent Advances and Future Outlooks
Abstract
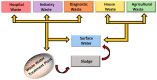
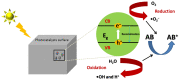
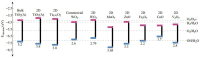
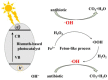
The existence of antibiotics in the environment can trigger a number of issues by fostering the widespread development of antimicrobial resistance. Currently, the most popular techniques for removing antibiotic pollutants from water include physical adsorption, flocculation, and chemical oxidation, however, these processes usually leave a significant quantity of chemical reagents and polymer electrolytes in the water, which can lead to difficulty post-treating unmanageable deposits. Furthermore, though cost-effectiveness, efficiency, reaction conditions, and nontoxicity during the degradation of antibiotics are hurdles to overcome, a variety of photocatalysts can be used to degrade pollutant residuals, allowing for a number of potential solutions to these issues. Thus, the urgent need for effective and rapid processes for photocatalytic degradation leads to an increased interest in finding more sustainable catalysts for antibiotic degradation. In this review, we provide an overview of the removal of pharmaceutical antibiotics through photocatalysis, and detail recent progress using different nanostructure-based photocatalysts. We also review the possible sources of antibiotic pollutants released through the ecological chain and the consequences and damages caused by antibiotics in wastewater on the environment and human health. The fundamental dynamic processes of nanomaterials and the degradation mechanisms of antibiotics are then discussed, and recent studies regarding different photocatalytic materials for the degradation of some typical and commonly used antibiotics are comprehensively summarized. Finally, major challenges and future opportunities for the photocatalytic degradation of commonly used antibiotics are highlighted.
Keywords: antibiotics; degradation mechanism; photocatalysts; photocatalytic degradation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Recent advances and future trends in metal oxide photocatalysts for removal of pharmaceutical pollutants from wastewater: a comprehensive review.Environ Geochem Health. 2024 Aug 10;46(9):364. doi: 10.1007/s10653-024-02140-x. Environ Geochem Health. 2024. PMID: 39126526 Review.
-
Advancements in iron-based photocatalytic degradation for antibiotics and dyes.J Environ Manage. 2025 Feb;374:123991. doi: 10.1016/j.jenvman.2024.123991. Epub 2025 Jan 14. J Environ Manage. 2025. PMID: 39813802 Review.
-
A review of tungsten trioxide (WO3)-based materials for antibiotics removal via photocatalysis.Ecotoxicol Environ Saf. 2023 Jul 1;259:114988. doi: 10.1016/j.ecoenv.2023.114988. Epub 2023 May 12. Ecotoxicol Environ Saf. 2023. PMID: 37182300 Review.
-
Removal of tetracycline from wastewater using g-C3N4 based photocatalysts: A review.Environ Res. 2023 Jan 1;216(Pt 3):114660. doi: 10.1016/j.envres.2022.114660. Epub 2022 Nov 8. Environ Res. 2023. PMID: 36368373 Review.
-
Recent advances based on the synergetic effect of adsorption for removal of dyes from waste water using photocatalytic process.J Environ Sci (China). 2018 Mar;65:201-222. doi: 10.1016/j.jes.2017.03.011. Epub 2017 Mar 17. J Environ Sci (China). 2018. PMID: 29548392 Review.
Cited by
-
Removal of Rifampicin and Rifaximin Antibiotics on PET Fibers: Optimization, Modeling, and Mechanism Insight.Polymers (Basel). 2025 Jul 30;17(15):2089. doi: 10.3390/polym17152089. Polymers (Basel). 2025. PMID: 40808138 Free PMC article.
-
The Microbial Assay for Risk Assessment (MARA) in the Assessment of the Antimicrobial Activity of Ofloxacin and Its Photoproducts.Int J Mol Sci. 2025 Mar 13;26(6):2595. doi: 10.3390/ijms26062595. Int J Mol Sci. 2025. PMID: 40141235 Free PMC article.
-
Characterization and Isolation of the Major Biologically Active Metabolites Isolated from Ficus retusa and Their Synergistic Effect with Tetracycline against Certain Pathogenic-Resistant Bacteria.Pharmaceuticals (Basel). 2022 Nov 26;15(12):1473. doi: 10.3390/ph15121473. Pharmaceuticals (Basel). 2022. PMID: 36558923 Free PMC article.
-
Light-driven photocatalysis as an effective tool for degradation of antibiotics.RSC Adv. 2024 Jun 27;14(29):20492-20515. doi: 10.1039/d4ra03431g. eCollection 2024 Jun 27. RSC Adv. 2024. PMID: 38946773 Free PMC article. Review.
-
Green synthesis of copper oxide nanoparticles and its efficiency in degradation of rifampicin antibiotic.Sci Rep. 2023 Aug 28;13(1):14030. doi: 10.1038/s41598-023-41119-z. Sci Rep. 2023. PMID: 37640783 Free PMC article.
References
-
- Abdurahman M.H., Abdullah A.Z., Shoparwe N.F. A Comprehensive Review on Sonocatalytic, Photocatalytic, and Sonophotocatalytic Processes for the Degradation of Antibiotics in Water: Synergistic Mechanism and Degradation Pathway. Chem. Eng. J. 2021;413:127412. doi: 10.1016/j.cej.2020.127412. - DOI
-
- Calvete M.J.F., Piccirillo G., Vinagreiro C.S., Pereira M.M. Hybrid Materials for Heterogeneous Photocatalytic Degradation of Antibiotics. Coord. Chem. Rev. 2019;395:63–85. doi: 10.1016/j.ccr.2019.05.004. - DOI
-
- Shurbaji S., Huong P.T., Altahtamouni T.M. Review on the Visible Light Photocatalysis for the Decomposition of Ciprofloxacin, Norfloxacin, Tetracyclines, and Sulfonamides Antibiotics in Wastewater. Catalysts. 2021;11:437. doi: 10.3390/catal11040437. - DOI
-
- Li D., Shi W. Recent Developments in Visible-Light Photocatalytic Degradation of Antibiotics. Chin. J. Catal. 2016;37:792–799. doi: 10.1016/S1872-2067(15)61054-3. - DOI
-
- Wei Z., Liu J., Shangguan W. A Review on Photocatalysis in Antibiotic Wastewater: Pollutant Degradation and Hydrogen Production. Chin. J. Catal. 2020;41:1440–1450. doi: 10.1016/S1872-2067(19)63448-0. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

