Mammalian Sulfatases: Biochemistry, Disease Manifestation, and Therapy
- PMID: 35897729
- PMCID: PMC9330403
- DOI: 10.3390/ijms23158153
Mammalian Sulfatases: Biochemistry, Disease Manifestation, and Therapy
Abstract
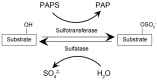
Sulfatases are enzymes that catalyze the removal of sulfate from biological substances, an essential process for the homeostasis of the body. They are commonly activated by the unusual amino acid formylglycine, which is formed from cysteine at the catalytic center, mediated by a formylglycine-generating enzyme as a post-translational modification. Sulfatases are expressed in various cellular compartments such as the lysosome, the endoplasmic reticulum, and the Golgi apparatus. The substrates of mammalian sulfatases are sulfolipids, glycosaminoglycans, and steroid hormones. These enzymes maintain neuronal function in both the central and the peripheral nervous system, chondrogenesis and cartilage in the connective tissue, detoxification from xenobiotics and pharmacological compounds in the liver, steroid hormone inactivation in the placenta, and the proper regulation of skin humidification. Human sulfatases comprise 17 genes, 10 of which are involved in congenital disorders, including lysosomal storage disorders, while the function of the remaining seven is still unclear. As for the genes responsible for pathogenesis, therapeutic strategies have been developed. Enzyme replacement therapy with recombinant enzyme agents and gene therapy with therapeutic transgenes delivered by viral vectors are administered to patients. In this review, the biochemical substrates, disease manifestation, and therapy for sulfatases are summarized.
Keywords: biochemistry; formylglycine; gene therapy; post-translational modification; sulfatase.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Lysosomal sulfatases: a growing family.Biochem J. 2020 Oct 30;477(20):3963-3983. doi: 10.1042/BCJ20200586. Biochem J. 2020. PMID: 33120425 Review.
-
A novel protein modification generating an aldehyde group in sulfatases: its role in catalysis and disease.Bioessays. 1998 Jun;20(6):505-10. doi: 10.1002/(SICI)1521-1878(199806)20:6<505::AID-BIES9>3.0.CO;2-K. Bioessays. 1998. PMID: 9699462 Review.
-
SUMF1 enhances sulfatase activities in vivo in five sulfatase deficiencies.Biochem J. 2007 Apr 15;403(2):305-12. doi: 10.1042/BJ20061783. Biochem J. 2007. PMID: 17206939 Free PMC article.
-
Sulfatase activities towards the regulation of cell metabolism and signaling in mammals.Cell Mol Life Sci. 2010 Mar;67(5):769-80. doi: 10.1007/s00018-009-0203-3. Epub 2009 Nov 22. Cell Mol Life Sci. 2010. PMID: 20165970 Free PMC article. Review.
-
Molecular basis of multiple sulfatase deficiency, mucolipidosis II/III and Niemann-Pick C1 disease - Lysosomal storage disorders caused by defects of non-lysosomal proteins.Biochim Biophys Acta. 2009 Apr;1793(4):710-25. doi: 10.1016/j.bbamcr.2008.11.015. Epub 2008 Dec 10. Biochim Biophys Acta. 2009. PMID: 19124046 Review.
Cited by
-
New Insights into the Plutella xylostella Detoxifying Enzymes: Sequence Evolution, Structural Similarity, Functional Diversity, and Application Prospects of Glucosinolate Sulfatases.J Agric Food Chem. 2023 Jul 26;71(29):10952-10969. doi: 10.1021/acs.jafc.3c03246. Epub 2023 Jul 18. J Agric Food Chem. 2023. PMID: 37462091 Free PMC article. Review.
-
Promiscuous Enzymes for Residue-Specific Peptide and Protein Late-Stage Functionalization.Chembiochem. 2023 Sep 1;24(17):e202300372. doi: 10.1002/cbic.202300372. Epub 2023 Jul 19. Chembiochem. 2023. PMID: 37338668 Free PMC article. Review.
-
Discovery and development of steroidal enzyme inhibitors as anti-cancer drugs: state-of-the-art and future perspectives.J Enzyme Inhib Med Chem. 2025 Dec;40(1):2483818. doi: 10.1080/14756366.2025.2483818. Epub 2025 Apr 2. J Enzyme Inhib Med Chem. 2025. PMID: 40172115 Free PMC article. Review.
-
Steroid sulfatase and sulfotransferases in the estrogen and androgen action of gynecological cancers: current status and perspectives.Essays Biochem. 2024 Dec 4;68(4):411-422. doi: 10.1042/EBC20230096. Essays Biochem. 2024. PMID: 38994718 Free PMC article. Review.
-
Arylsulfatase K attenuates airway epithelial cell senescence in COPD by regulating parkin-mediated mitophagy.Redox Biol. 2025 Jul 31;86:103793. doi: 10.1016/j.redox.2025.103793. Online ahead of print. Redox Biol. 2025. PMID: 40763656 Free PMC article.
References
-
- Barbeyron T., Brillet-Guéguen L., Carré W., Carrière C., Caron C., Czjzek M., Hoebeke M., Michel G. Matching the Diversity of Sulfated Biomolecules: Creation of a Classification Database for Sulfatases Reflecting Their Substrate Specificity. PLoS ONE. 2016;11:e0164846. doi: 10.1371/journal.pone.0164846. - DOI - PMC - PubMed
-
- Biffi A., Capotondo A., Fasano S., del Carro U., Marchesini S., Azuma H., Malaguti M.C., Amadio S., Brambilla R., Grompe M., et al. Gene Therapy of Metachromatic Leukodystrophy Reverses Neurological Damage and Deficits in Mice. J. Clin. Investig. 2006;116:3070–3082. doi: 10.1172/JCI28873. - DOI - PMC - PubMed
-
- Hendrickx G., Danyukova T., Baranowsky A., Rolvien T., Angermann A., Schweizer M., Keller J., Schröder J., Meyer-Schwesinger C., Muschol N., et al. Enzyme Replacement Therapy in Mice Lacking Arylsulfatase B Targets Bone-Remodeling Cells, but not Chondrocytes. Hum. Mol. Genet. 2020;29:803–816. doi: 10.1093/hmg/ddaa006. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

