Simultaneous Dual Recordings From Vestibular Hair Cells and Their Calyx Afferents Demonstrate Multiple Modes of Transmission at These Specialized Endings
- PMID: 35899268
- PMCID: PMC9310783
- DOI: 10.3389/fneur.2022.891536
Simultaneous Dual Recordings From Vestibular Hair Cells and Their Calyx Afferents Demonstrate Multiple Modes of Transmission at These Specialized Endings
Abstract
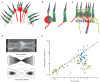
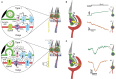
In the vestibular periphery, transmission via conventional synaptic boutons is supplemented by post-synaptic calyceal endings surrounding Type I hair cells. This review focusses on the multiple modes of communication between these receptors and their enveloping calyces as revealed by simultaneous dual-electrode recordings. Classic orthodromic transmission is accompanied by two forms of bidirectional communication enabled by the extensive cleft between the Type I hair cell and its calyx. The slowest cellular communication low-pass filters the transduction current with a time constant of 10-100 ms: potassium ions accumulate in the synaptic cleft, depolarizing both the hair cell and afferent to potentials greater than necessary for rapid vesicle fusion in the receptor and potentially triggering action potentials in the afferent. On the millisecond timescale, conventional glutamatergic quantal transmission occurs when hair cells are depolarized to potentials sufficient for calcium influx and vesicle fusion. Depolarization also permits a third form of transmission that occurs over tens of microseconds, resulting from the large voltage- and ion-sensitive cleft-facing conductances in both the hair cell and the calyx that are open at their resting potentials. Current flowing out of either the hair cell or the afferent divides into the fraction flowing across the cleft into its cellular partner, and the remainder flowing out of the cleft and into the surrounding fluid compartment. These findings suggest multiple biophysical bases for the extensive repertoire of response dynamics seen in the population of primary vestibular afferent fibers. The results further suggest that evolutionary pressures drive selection for the calyx afferent.
Keywords: calyx; ephaptic transmission; hair cell; ion accumulation; quantal transmission; resistive coupling; synaptic transmission; vestibular.
Copyright © 2022 Contini, Holstein and Art.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Simultaneous recordings from vestibular Type I hair cells and their calyceal afferents in mice.Front Neurol. 2024 Aug 27;15:1434026. doi: 10.3389/fneur.2024.1434026. eCollection 2024. Front Neurol. 2024. PMID: 39263277 Free PMC article.
-
Accumulation of K+ in the synaptic cleft modulates activity by influencing both vestibular hair cell and calyx afferent in the turtle.J Physiol. 2017 Feb 1;595(3):777-803. doi: 10.1113/JP273060. Epub 2016 Nov 4. J Physiol. 2017. PMID: 27633787 Free PMC article.
-
Synaptic cleft microenvironment influences potassium permeation and synaptic transmission in hair cells surrounded by calyx afferents in the turtle.J Physiol. 2020 Feb;598(4):853-889. doi: 10.1113/JP278680. Epub 2019 Nov 29. J Physiol. 2020. PMID: 31623011 Free PMC article.
-
Synaptic transmission at the vestibular hair cells of amniotes.Mol Cell Neurosci. 2022 Jul;121:103749. doi: 10.1016/j.mcn.2022.103749. Epub 2022 Jun 3. Mol Cell Neurosci. 2022. PMID: 35667549 Review.
-
Morphophysiological and ultrastructural studies in the mammalian cristae ampullares.Hear Res. 1990 Nov;49(1-3):89-102. doi: 10.1016/0378-5955(90)90097-9. Hear Res. 1990. PMID: 2292511 Review.
Cited by
-
Anatomical and molecular insights into avian inner ear sensory hair cell regeneration.Dev Biol. 2025 Sep;525:13-25. doi: 10.1016/j.ydbio.2025.05.021. Epub 2025 May 23. Dev Biol. 2025. PMID: 40414451 Review.
-
Simultaneous recordings from vestibular Type I hair cells and their calyceal afferents in mice.Front Neurol. 2024 Aug 27;15:1434026. doi: 10.3389/fneur.2024.1434026. eCollection 2024. Front Neurol. 2024. PMID: 39263277 Free PMC article.
-
Vestibular afferent neurons develop normally in the absence of quantal/glutamatergic input.Front Neurol. 2024 Nov 25;15:1441964. doi: 10.3389/fneur.2024.1441964. eCollection 2024. Front Neurol. 2024. PMID: 39655160 Free PMC article.
-
Using macular velocity measurements to relate parameters of bone conduction to vestibular compound action potential responses.Sci Rep. 2023 Jun 23;13(1):10204. doi: 10.1038/s41598-023-37102-3. Sci Rep. 2023. PMID: 37353559 Free PMC article.
-
The neural basis of vestibular evoked myogenic potentials. The cVEMP is a specific indicator of saccular function.Front Neurol. 2025 Aug 6;16:1644120. doi: 10.3389/fneur.2025.1644120. eCollection 2025. Front Neurol. 2025. PMID: 40843260 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

