Behavioral studies of spinal conditioning: The spinal cord is smarter than you think it is
- PMID: 35901417
- PMCID: PMC10391333
- DOI: 10.1037/xan0000332
Behavioral studies of spinal conditioning: The spinal cord is smarter than you think it is
Abstract
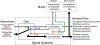
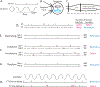
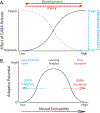
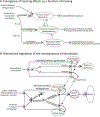
In 1988 Robert Rescorla published an article in the Annual Review of Neuroscience that addressed the circumstances under which learning occurs, some key methodological issues, and what constitutes an example of learning. The article has inspired a generation of neuroscientists, opening the door to a wider range of learning phenomena. After reviewing the historical context for his article, its key points are briefly reviewed. The perspective outlined enabled the study of learning in simpler preparations, such as the spinal cord. The period after 1988 revealed that pain (nociceptive) stimuli can induce a lasting sensitization of spinal cord circuits, laying down a kind of memory mediated by signal pathways analogous to those implicated in brain dependent learning and memory. Evidence suggests that the spinal cord is sensitive to instrumental response-outcome (R-O) relations, that learning can induce a peripheral modification (muscle memory) that helps maintain the learned response, and that learning can promote adaptive plasticity (a form of metaplasticity). Conversely, exposure to uncontrollable stimulation disables the capacity to learn. Spinal cord neurons can also abstract that stimuli occur in a regular (predictable) manner, a capacity that appears linked to a neural oscillator (central pattern generator). Disrupting communication with the brain has been shown to transform how GABA affects neuronal function (an example of ionic plasticity), releasing a brake that enables plasticity. We conclude by presenting a framework for understanding these findings and the implications for the broader study of learning. (PsycInfo Database Record (c) 2022 APA, all rights reserved).
Conflict of interest statement
Figures


Similar articles
-
Metaplasticity within the spinal cord: Evidence brain-derived neurotrophic factor (BDNF), tumor necrosis factor (TNF), and alterations in GABA function (ionic plasticity) modulate pain and the capacity to learn.Neurobiol Learn Mem. 2018 Oct;154:121-135. doi: 10.1016/j.nlm.2018.04.007. Epub 2018 Apr 7. Neurobiol Learn Mem. 2018. PMID: 29635030 Free PMC article.
-
Metaplasticity and behavior: how training and inflammation affect plastic potential within the spinal cord and recovery after injury.Front Neural Circuits. 2014 Sep 8;8:100. doi: 10.3389/fncir.2014.00100. eCollection 2014. Front Neural Circuits. 2014. PMID: 25249941 Free PMC article. Review.
-
Learning from the spinal cord: how the study of spinal cord plasticity informs our view of learning.Neurobiol Learn Mem. 2014 Feb;108:155-71. doi: 10.1016/j.nlm.2013.08.003. Epub 2013 Aug 20. Neurobiol Learn Mem. 2014. PMID: 23973905 Free PMC article. Review.
-
Learning to promote recovery after spinal cord injury.Exp Neurol. 2020 Aug;330:113334. doi: 10.1016/j.expneurol.2020.113334. Epub 2020 Apr 28. Exp Neurol. 2020. PMID: 32353465 Free PMC article. Review.
-
Ionic plasticity and pain: The loss of descending serotonergic fibers after spinal cord injury transforms how GABA affects pain.Exp Neurol. 2018 Aug;306:105-116. doi: 10.1016/j.expneurol.2018.05.002. Epub 2018 May 2. Exp Neurol. 2018. PMID: 29729247 Free PMC article.
Cited by
-
Updating perspectives on spinal cord function: motor coordination, timing, relational processing, and memory below the brain.Front Syst Neurosci. 2024 Feb 20;18:1184597. doi: 10.3389/fnsys.2024.1184597. eCollection 2024. Front Syst Neurosci. 2024. PMID: 38444825 Free PMC article. Review.
-
Ionic Plasticity: Common Mechanistic Underpinnings of Pathology in Spinal Cord Injury and the Brain.Cells. 2022 Sep 17;11(18):2910. doi: 10.3390/cells11182910. Cells. 2022. PMID: 36139484 Free PMC article. Review.
References
-
- Abraham WC, & Bear MF (1996). Metaplasticity: the plasticity of synaptic plasticity. Trends Neurosci, 19(4), 126–130. - PubMed
-
- Akil H, Watson S, Young E, Lewis ME, Khachaturiam H, & Walker JM (1984). Endogenous opioids: Biology and function. Annual Review of Neuroscience, 7, 223–255. - PubMed
-
- Alkon DL (1983). Learning in a marine snail. Scientific American, 249(1), 70–74. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

