Pilot randomized controlled trial evaluating the effect of random nicotine delivery on cigarettes per day and smoke exposure
- PMID: 35901983
- PMCID: PMC9743484
- DOI: 10.1016/j.brainresbull.2022.07.017
Pilot randomized controlled trial evaluating the effect of random nicotine delivery on cigarettes per day and smoke exposure
Abstract
Background: Many smokers report attempting to quit each year, yet most relapse, in part due to exposure to smoking-related cues. It is hypothesized that extinction of the cue-drug association could be facilitated through random nicotine delivery (RND), thus making it easier for smokers to quit. The current study aimed to evaluate the effects of RND on smoking cessation-related outcomes including cigarettes per day (CPD) and exhaled carbon monoxide (CO).
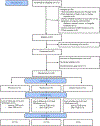
Methods: Participants were current smokers (>9 CPD) interested in quitting. Novel trans-mucosal, orally dissolving nicotine films, developed by Bionex Pharmaceuticals, were used in the study. The pharmacokinetic profile of these films was assessed in single (Experiment 1) and multiple-dose (Experiment 2) administrations prior to the smoking cessation study (Experiment 3). In Experiment 3, participants were randomized 1:1:1 to recieve 4 nicotine films per day of either: placebo delivery (0 mg), steady-state delivery (2 mg), or random nicotine delivery (RND) (0 mg or 4 mg). After two weeks, participants were advised to quit (target quit date, TQD) and were followed up 4 weeks later to collect CPD and CO and to measure dependence (Penn State Cigarette Dependence Index; PSCDI) and craving (Questionnaire of Smoking Urges; QSU-Brief). Means and frequencies were used to describe the data and repeated measures ANOVA was used to determine differences between groups.
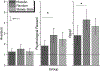
Results: The pharmacokinetic studies (Experiment 1 and 2) demonstrated that the films designed for this study delivered nicotine as expected, with the 4 mg film delivering a nicotine boost of approximately 12.4 ng/mL across both the single and the multiple dose administration studies. The films reduced craving for a cigarette and were well-tolerated, overall, and caused no changes in blood pressure or heart rate. Using these films in the cessation study (Experiment 3) (n = 45), there was a significant overall reduction in cigarettes smoked per day (CPD) and in exhaled CO, with no significant differences across groups (placebo, steady-state, RND). In addition, there were no group differences in dependence or craving. Adverse events included heartburn, hiccups, nausea, and to a lesser extent, vomiting and anxiety and there were no differences across groups.
Conclusion: Overall, this pilot study found that RND via orally dissolving films was feasible and well tolerated by participants. However, RND participants did not experience a greater reduction in self-reported CPD and exhaled CO, compared with participants in the steady-state and placebo delivery groups. Future studies to evaluate optimal RND parameters with larger sample sizes are needed to fully understand the effect of RND on smoking cessation-related outcomes.
Keywords: Nicotine delivery; Nicotine film; Nicotine replacement therapy; Pharmacokinetics.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest
JF has done paid consulting for pharmaceutical companies involved in producing smoking cessation medications, including GSK, Pfizer, Novartis, J&J, and Cypress Bioscience. HST is the Director of Ventures & Technologies at Bionex Pharmaceuticals LLC. The other authors have no disclosures to report related to this publication.
Figures
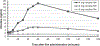


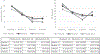
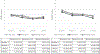

References
-
- Babb S, Malarcher A, Schauer G, Asman K, Jamal A, 2017. Quitting smoking among adults - United States, 2000–2015. Morb. Mortal. Wkly. Rep. 65 (52), 1457–1464. - PubMed
-
- Belita E, Sidani S, 2015. Attrition in smoking cessation intervention studies: a systematic review. Can. J. Nurs. Res. = Rev. Can. De. Rech. En. Sci. Infirm. 47 (4), 21–40. - PubMed
-
- Choi JH, Dresler CM, Norton MR, Strahs KR, 2003. Pharmacokinetics of a nicotine polacrilex lozenge. Nicot. Tob. Res. 5 (5), 635–644. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

