Monosodium iodoacetate-induced subchondral bone microstructure and inflammatory changes in an animal model of osteoarthritis
- PMID: 35903181
- PMCID: PMC9287849
- DOI: 10.1515/biol-2022-0079
Monosodium iodoacetate-induced subchondral bone microstructure and inflammatory changes in an animal model of osteoarthritis
Abstract
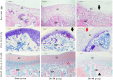
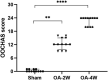
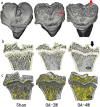
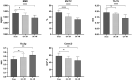
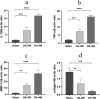
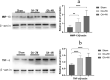
The monosodium iodoacetate (MIA)-induced osteoarthritis (OA) may lead to cartilage degeneration and histopathological lesions. However, the correlation between inflammatory reaction and subchondral bone remodeling in a rodent osteoarthritic model is ambiguous. In this study, intra-articular injection of MIA was performed in 36 four-week-old specific pathogen-free male Wistar rats to induce OA. After 4 weeks of intervention, changes in intrinsic structural properties of the subchondral bones were measured, and the histological evaluation, as well as biochemical analysis, was conducted. We found that intra-articular injection of MIA increased chondrocyte apoptosis and promoted cartilage matrix degradation, such as cartilage surface defects and shallow or disappearing staining. MIA also induced inflammation, improved the expression of IL-1β, TNF-α, and matrix metalloproteinase, and decreased the expression of cartilage-specific proteins with the extension of modeling time. Meanwhile, the MIA also significantly accelerated the subchondral bone remodeling, as shown by the decreased subchondral bone density, thinning of trabeculae, disordered cartilage structure, and morphology. In conclusion, we have shown that MIA-induced rodent osteoarthritic model would cause decreased subchondral bone density, sparse trabecular bone, and other manifestations of osteoporosis accompanied by an inflammatory response, which would worsen with the progression of modeling time. Our results suggest that different phases of MIA-induced OA are associated with the changes in subchondral bone microstructure and the progression of local inflammation.
Keywords: animal model; bone microarchitecture; iodoacetic acid; osteoarthritis.
© 2022 Zheming Bao et al., published by De Gruyter.
Conflict of interest statement
Conflict of interest: Authors state no conflict of interest.
Figures

Similar articles
-
Application of in vivo micro-computed tomography in the temporal characterisation of subchondral bone architecture in a rat model of low-dose monosodium iodoacetate-induced osteoarthritis.Arthritis Res Ther. 2011;13(6):R210. doi: 10.1186/ar3543. Epub 2011 Dec 21. Arthritis Res Ther. 2011. PMID: 22185204 Free PMC article.
-
Effect of interval-training exercise on subchondral bone in a chemically-induced osteoarthritis model.Osteoarthritis Cartilage. 2014 Aug;22(8):1176-85. doi: 10.1016/j.joca.2014.05.020. Epub 2014 Jun 10. Osteoarthritis Cartilage. 2014. PMID: 24928318
-
Pulsed electromagnetic field at different stages of knee osteoarthritis in rats induced by low-dose monosodium iodoacetate: Effect on subchondral trabecular bone microarchitecture and cartilage degradation.Bioelectromagnetics. 2017 Apr;38(3):227-238. doi: 10.1002/bem.22028. Epub 2016 Dec 27. Bioelectromagnetics. 2017. PMID: 28026095
-
Pre-emptive, early, and delayed alendronate treatment in a rat model of knee osteoarthritis: effect on subchondral trabecular bone microarchitecture and cartilage degradation of the tibia, bone/cartilage turnover, and joint discomfort.Osteoarthritis Cartilage. 2013 Oct;21(10):1595-604. doi: 10.1016/j.joca.2013.06.020. Epub 2013 Jul 1. Osteoarthritis Cartilage. 2013. PMID: 23827368
-
The Pharmacology of Pain Associated With the Monoiodoacetate Model of Osteoarthritis.Front Pharmacol. 2019 Sep 18;10:974. doi: 10.3389/fphar.2019.00974. eCollection 2019. Front Pharmacol. 2019. PMID: 31619987 Free PMC article. Review.
Cited by
-
Advancing Osteoarthritis Research: Insights from Rodent Models and Emerging Trends.J Orthop Sports Med. 2025;7(1):110-128. doi: 10.26502/josm.511500187. Epub 2025 Mar 14. J Orthop Sports Med. 2025. PMID: 40264810 Free PMC article.
-
Artemisinin emulgel ameliorates cartilage degradation in knee osteoarthritis: in vitro and in vivo studies.Ther Deliv. 2024;15(12):939-955. doi: 10.1080/20415990.2024.2418281. Epub 2024 Nov 6. Ther Deliv. 2024. PMID: 39503537
-
Mitochondria-Targeted Cassic Acid Base Nanoprodrug for Enhanced Anti-Inflammatory and Osteogenic Effects in Osteoarthritis.ACS Omega. 2025 Aug 11;10(32):35940-35953. doi: 10.1021/acsomega.5c03216. eCollection 2025 Aug 19. ACS Omega. 2025. PMID: 40852312 Free PMC article.
-
Polyherbal formulation PL02 alleviates pain, inflammation, and subchondral bone deterioration in an osteoarthritis rodent model.Front Nutr. 2023 Nov 16;10:1217051. doi: 10.3389/fnut.2023.1217051. eCollection 2023. Front Nutr. 2023. PMID: 38045809 Free PMC article.
References
-
- Chin K-Y, Wong SK, Japar Sidik FZ, Abdul Hamid J, Abas NH, Mohd Ramli ES, et al. The effects of annatto tocotrienol supplementation on cartilage and subchondral bone in an animal model of osteoarthritis induced by monosodium iodoacetate. Int J Env Res Pub He. 2019 Aug 13;16(16):2897. 10.3390/ijerph16162897. - DOI - PMC - PubMed
-
- Lampropoulou-Adamidou K, Lelovas P, Karadimas EV, Liakou C, Triantafillopoulos IK, Dontas I, et al. Useful animal models for the research of osteoarthritis. Eur J Orthop Surg Traumatol. 2014 Apr;24(3):263–71. - PubMed
LinkOut - more resources
Full Text Sources
