Stem cell transplantation for systemic sclerosis
- PMID: 35904231
- PMCID: PMC9336163
- DOI: 10.1002/14651858.CD011819.pub2
Stem cell transplantation for systemic sclerosis
Abstract
Background: Systemic sclerosis (SSc) is a chronic autoimmune disease characterized by systemic inflammation, fibrosis, vascular injury, reduced quality of life, and limited treatment options. Autologous hematopoietic stem cell transplantation (HSCT) has emerged as a potential intervention for severe SSc refractory to conventional treatment.
Objectives: To assess the benefits and harms of autologous hematopoietic stem cell transplantation for the treatment of systemic sclerosis (specifically, non-selective myeloablative HSCT versus cyclophosphamide; selective myeloablative HSCT versus cyclophosphamide; non-selective non-myeloablative HSCT versus cyclophosphamide).
Search methods: We searched for randomized controlled trials (RCTs) in CENTRAL, MEDLINE, Embase, and trial registries from database insertion to 4 February 2022.
Selection criteria: We included RCTs that compared HSCT to immunomodulators in the treatment of SSc.
Data collection and analysis: Two review authors independently selected studies for inclusion, extracted study data, and performed risk of bias and GRADE assessments to assess the certainty of evidence using standard Cochrane methods.
Main results: We included three RCTs evaluating: non-myeloablative non-selective HSCT (10 participants), non-myeloablative selective HSCT (79 participants), and myeloablative selective HSCT (36 participants). The comparator in all studies was cyclophosphamide (123 participants). The study examining non-myeloablative non-selective HSCT had a high risk of bias given the differences in baseline characteristics between the two arms. The other studies had a high risk of detection bias for participant-reported outcomes. The studies had follow-up periods of one to 4.5 years. Most participants had severe disease, mean age 40 years, and the duration of disease was less than three years. Efficacy No study demonstrated an overall mortality benefit of HSCT when compared to cyclophosphamide. However, non-myeloablative selective HSCT showed overall survival benefits using Kaplan-Meier curves at 10 years and myeloablative selective HSCT at six years. We graded our certainty of evidence as moderate for non-myeloablative selective HSCT and myeloablative selective HSCT. Certainty of evidence was low for non-myeloablative non-selective HSCT. Event-free survival was improved compared to cyclophosphamide with non-myeloablative selective HSCT at 48 months (hazard ratio (HR) 0.34, 95% confidence interval (CI) 0.16 to 0.74; moderate-certainty evidence). There was no improvement with myeloablative selective HSCT at 54 months (HR 0.54 95% CI 0.23 to 1.27; moderate-certainty evidence). The non-myeloablative non-selective HSCT trial did not report event-free survival. There was improvement in functional ability measured by the Health Assessment Questionnaire Disability Index (HAQ-DI, scale from 0 to 3 with 3 being very severe functional impairment) with non-myeloablative selective HSCT after two years with a mean difference (MD) of -0.39 (95% CI -0.72 to -0.06; absolute treatment benefit (ATB) -13%, 95% CI -24% to -2%; relative percent change (RPC) -27%, 95% CI -50% to -4%; low-certainty evidence). Myeloablative selective HSCT demonstrated a risk ratio (RR) for improvement of 3.4 at 54 months (95% CI 1.5 to 7.6; ATB -37%, 95% CI -18% to -57%; RPC -243%, 95% CI -54% to -662%; number needed to treat for an additional beneficial outcome (NNTB) 3, 95% CI 2 to 9; low-certainty evidence). The non-myeloablative non-selective HSCT trial did not report HAQ-DI results. All transplant modalities showed improvement of modified Rodnan skin score (mRSS) (scale from 0 to 51 with the higher number being more severe skin thickness) favoring HSCT over cyclophosphamide. At two years, non-myeloablative selective HSCT showed an MD in mRSS of -11.1 (95% CI -14.9 to -7.3; ATB -22%, 95% CI -29% to -14%; RPC -43%, 95% CI -58% to -28%; moderate-certainty evidence). At 54 months, myeloablative selective HSCT at showed a greater improvement in skin scores than the cyclophosphamide group (RR 1.51, 95% CI 1.06 to 2.13; ATB -27%, 95% CI -6% to -47%; RPC -51%, 95% CI -6% to -113%; moderate-certainty evidence). The NNTB was 4 (95% CI 3 to 18). At one year, for non-myeloablative non-selective HSCT the MD was -16.00 (95% CI -26.5 to -5.5; ATB -31%, 95% CI -52% to -11%; RPC -84%, 95% CI -139% to -29%; low-certainty evidence). No studies reported data on pulmonary arterial hypertension. Adverse events In the non-myeloablative selective HSCT study, there were 51/79 serious adverse events with HSCT and 30/77 with cyclophosphamide (RR 1.7, 95% CI 1.2 to 2.3), with an absolute risk increase of 26% (95% CI 10% to 41%), and a relative percent increase of 66% (95% CI 20% to 129%). The number needed to treat for an additional harmful outcome was 4 (95% CI 3 to 11) (moderate-certainty evidence). In the myeloablative selective HSCT study, there were similar rates of serious adverse events between groups (25/34 with HSCT and 19/37 with cyclophosphamide; RR 1.43, 95% CI 0.99 to 2.08; moderate-certainty evidence). The non-myeloablative non-selective HSCT trial did not clearly report serious adverse events.
Authors' conclusions: Non-myeloablative selective and myeloablative selective HSCT had moderate-certainty evidence for improvement in event-free survival, and skin thicknesscompared to cyclophosphamide. There is also low-certainty evidence that these modalities of HSCT improve physical function. However, non-myeloablative selective HSCT and myeloablative selective HSCT resulted in more serious adverse events than cyclophosphamide; highlighting the need for careful risk-benefit considerations for people considering these HSCTs. Evidence for the efficacy and adverse effects of non-myeloablative non-selective HSCT is limited at this time. Due to evidence provided from one study with high risk of bias, we have low-certainty evidence that non-myeloablative non-selective HSCT improves outcomes in skin scores, forced vital capacity, and safety. Two modalities of HSCT appeared to be a promising treatment option for SSc though there is a high risk of early treatment-related mortality and other adverse events. Additional research is needed to determine the effectiveness and adverse effects of non-myeloablative non-selective HSCT in the treatment of SSc. Also, more studies will be needed to determine how HSCT compares to other treatment options such as mycophenolate mofetil, as cyclophosphamide is no longer the first-line treatment for SSc. Finally, there is a need for a greater understanding of the role of HSCT for people with SSc with significant comorbidities or complications from SSc that were excluded from the trial criteria.
Trial registration: ClinicalTrials.gov NCT00278525 NCT00114530.
Copyright © 2022 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Conflict of interest statement
SB: none.
HS: none.
DAM: none.
MDM declares the following: Galapagos NV (Independent Contractor ‐ Consultant), Actelion Pharmaceuticals US, Inc. (Independent Contractor ‐ Consultant), Eicos Sciences (Independent Contractor ‐ Consultant), Boehringer Ingelheim (Independent Contractor ‐ Consultant)
KK: none.
MAL: none.
Figures
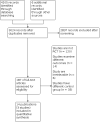






























Update of
References
References to studies included in this review
Burt 2011 {published data only}
-
- Burt RK, Shah SJ, Dill K, Grant T, Gheorghiade M, Schroeder J, et al. Autologous non-myeloablative haemopoietic stem-cell transplantations compared with pulse cyclophosphamide once per months for systemic sclerosis (ASSIST): an open-label, randomised phase 2 trial. Lancet 2011;378:498-506. - PubMed
-
- NCT00278525. Cyclophosphamide and rATG with hematopoietic stem cell support in systemic scleroderma. clinicaltrials.gov/ct2/show/NCT00278525 (first received 18 January 2006). [NCT00278525]
-
- NCT01445821. Autologous stem cell systemic sclerosis immune suppression trial (DIScl2011). clinicaltrials.gov/ct2/show/NCT01445821 (first received 4 October 2011). [NCT01445821]
Sullivan 2018 {published data only}
-
- Assassi S, Mayes MD, Pedroza C, Chang JT, Furst DE, Crofford LJ, et al. Immunoablation followed by autologous stem cell transplantation in systemic sclerosis patients decreases significantly the interferon signature. 2015 ACR/ARHP Annual Meeting; 6-11 Nov 2015; San Francisco (CA).
-
- Assassi S, Wallace P, Ying J, Keyes-Elstein L, Goldmuntz E, Turner J. Treatment with myeloablation followed by autologous stem cell transplantation normalizes the systemic sclerosis molecular signature. Journal of Scleroderma and Related Disorders 2018;3:s272-3.
-
- Assassi S, Wang X, Keyes-Elstein L, Goldmuntz E, Turner J, Zheng W. Changes in the systemic sclerosis molecular signatures after myeloablation followed by autologous hematopoietic stem cell transplantation and their clinical correlates. Arthritis and Rheumatology 2018;70(Suppl 9):s1002-3.
-
- Bellocchi C, Ying J, Goldmuntz E, Keyes-Elstein L, Varga J, Hinchcliff M, et al. Systemic sclerosis has a distinct serum protein profile that correlates with its clinical manifestations. Arthritis and Rheumatology 2018;70:s1213-4.
-
- Bellochi C, Sullivan K, Goldmuntz E, Ying J, Keyes-Elstein L, Varga J, et al. Myeloablation followed by autologous stem cell rescue leads to normalisation of serum IL-6 levels in patients with systemic sclerosis. Annals of the Rheumatic Diseases 2018;77:s1100-1.
van Laar 2014 {published data only (unpublished sought but not used)}ISRCTN54371254
-
- Abdallah NA, Wang M, Lansiaux P, Puyade M, Berthier S, Terriou L, et al. Long term outcomes of the French ASTIS systemic sclerosis cohort using the global rank composite score. Bone Marrow Transplantation 2021;56:2259-67. - PubMed
-
- Laar J, Farge D, Sont J and the EBMT/EULAR Scleroderma Study Group. LB0002. The ASTIS trial: autologous stem cell transplantation versus IV pulse cyclophosphamide in poor prognosis systemic sclerosis, first results. Annals of the Rheumatic Diseases 2013;71:151.
-
- Laar JM, Farge D, Sont J, Naraghi K, Marjanovic Z, Larghero J, et al. Autologous hematopoietic stem cell transplantation vs intravenous pulse cyclophosphamide in diffuse cutaneous systemic sclerosis. JAMA 2014;311(24):2490-8. - PubMed
-
- Laar JM, Farge D, Sont JK, Naraghi K, Marjanovic Z, Schuerwegh A, et al, EBMT/EULAR Scleroderma Study Group. High dose immunoablation and autologous hematopoietic stem cell transplantation versus monthly intravenous pulse therapy cyclophosphamide in severe systemic sclerosis. Arthritis & Rheumatism 2012;64(S10):4167.
References to ongoing studies
van Laar 2020 {unpublished data only}
-
- NCT04464434. Upfront autologous HSCT versus immunosuppression in early diffuse cutaneous systemic sclerosis (UPSIDE). clinicaltrials.gov/ct2/show/NCT04464434 (first received 9 July 2020).
Additional references
ACR 1980
-
- American College of Rheumatology. Preliminary criteria for the classification of systemic sclerosis (scleroderma). Subcommittee for scleroderma criteria of the American Rheumatism Association Diagnostic and Therapeutic Criteria Committee. Arthritis and Rheumatism 1980;23(5):581-90. [PMID: ] - PubMed
Bergamasco 2019
Binks 2001
Cates 2008 [Computer program]
-
- Visual Rx 2.0 NNT calculator. Cates C. Dr Chris Cates EBM, 2008. Available at www.nntonline.net.
Deeks 2021
-
- Deeks JJ, Higgins JP, Altman DG. Chapter 10: Analysing data and undertaking meta-analyses. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Distler 2008
-
- Distler O, Behrens F, Pittrow D, Huscher D, Denton CP, Foeldvari I, et al, EPOSS-Omeract Group. Defining appropriate outcome measures in pulmonary arterial hypertension related to systemic sclerosis: a Delphi consensus study with cluster analysis. Arthritis and Rheumatism 2008;59(6):867-75. [PMID: ] - PubMed
Elhai 2012
-
- Elhai M, Meune C, Avouac J, Kahan A, Allanore Y. Trends in mortality in patients with systemic sclerosis over 40 years: a systematic review and meta-analysis of cohort studies. Rheumatology (Oxford) 2012;51(6):1017-26. [PMID: ] - PubMed
EndNote X9 [Computer program]
-
- EndNote X9. Philadelphia (PA): Clarivate Analytics, 2019. Available at endnote.com.
Farge 2002
-
- Farge D, Marolleau JP, Zohar S, Marjanovic Z, Cabane J, Mounier N, et al, Intensification et Autogreffe dans les Maladies Auto Immunes Resistantes (ISAMAIR) Study Group. Autologous bone marrow transplantation in the treatment of refractory systemic sclerosis: early results from a French Multicentre phase I-II study. British Journal of Haematology 2002;119(3):726-39. [PMID: ] - PubMed
GRADEpro GDT [Computer program]
-
- GRADEpro GDT. Hamilton (ON): McMaster University (developed by Evidence Prime), accessed 3 March 2020. Available at gradepro.org.
Higgins 2017
-
- Higgins JP, Altman DG, Sterne JA. Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Churchill R, Chandler J, Cumpston MS, editor(s), Cochrane Handbook for Systematic Reviews of Interventions Version 5.2.0 (updated June 2017), The Cochrane Collaboration, 2017. Available from training.cochrane.org/handbook/archive/v5.2/.
Higgins 2021
-
- Higgins JP, Li T, Deeks JJ. Chapter 6: Choosing effect measures and computing estimates of effect. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Host 2017
-
- Host L, Nikpour M, Calderone A, Cannell P, Roddy J. Autologous stem cell transplantation in systemic sclerosis: a systematic review. Clinical and Experimental Rheumatology 2017;35(106):198-207. - PubMed
Hudson 2014
-
- Hudson M, Fritzler MJ. Diagnostic criteria of systemic sclerosis. Journal of Autoimmunity 2014;48-49:38-41. [PMID: ] - PubMed
Jordan 2015
-
- Jordan S, Maurer B, Toniolo M, Michel B, Distler O. Performance of the new ACR/EULAR classification criteria for systemic sclerosis in clinical practice. Rheumatology (Oxford) 2015;54(8):1454-8. - PubMed
Kafaja 2017
Khanna 2009
-
- Khanna D, Distler O, Avouac J, Behrens F, Clements PJ, Denton C, et al, CRISS, EPOSS. Measures of response in clinical trials of systemic sclerosis: the Combined Response Index for Systemic Sclerosis (CRISS) and Outcome Measures in Pulmonary Arterial Hypertension related to Systemic Sclerosis (EPOSS). Journal of Rheumatology 2009;36(10):2356-61. [PMID: ] - PubMed
Khanna 2019
Kowal‐Bielecka 2009
-
- Kowal-Bielecka O, Landewé R, Avouac J, Chwiesko S, Miniati I, Czirjak L, et al, EUSTAR Co-Authors. EULAR recommendations for the treatment of systemic sclerosis: a report from the EULAR Scleroderma Trials and Research group (EUSTAR). Annals of the Rheumatic Diseases 2009;68(5):620-8. [PMID: ] - PubMed
LeRoy 1988
-
- LeRoy EC, Black C, Fleischmajer R, Jablonska S, Krieg T, Medsger TA Jr, et al. Scleroderma (systemic sclerosis): classification, subsets and pathogenesis. Journal of Rheumatology 1988;15(2):202-5. [PMID: ] - PubMed
Mayes 2003
-
- Mayes MD, Lacey JV Jr, Beebe-Dimmer J, Gillespie BW, Cooper B, Laing TJ, et al. Prevalence, incidence, survival, and disease characteristics of systemic sclerosis in a large US population. Arthritis and Rheumatology 2003;48(8):2246-55. [PMID: ] - PubMed
Merkel 2003
-
- Merkel PA, Clements PJ, Reveille JD, Suarez-Almazor ME, Valentini G, Furst DE. Current status of outcome measure development of clinical trials in systemic sclerosis. Report from OMERACT 6. Journal of Rheumatology 2003;30(7):1630-47. [PMID: ] - PubMed
Nagaraja 2015
Nash 2007
Oyama 2007
-
- Oyama Y, Barr WG, Statkute L, Corbridge T, Gonda EA, Jovanovic B, et al. Autologous non-myeloablative hematopoietic stem cell transplantation in patients with systemic sclerosis. Bone Marrow Transplantation 2007;40(6):549-55. [PMID: ] - PubMed
Page 2021
-
- Page MJ, Higgins JP, Sterne JA. Chapter 13: Assessing risk of bias due to missing results in a synthesis. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Park 2015
Poormoghim 2012
-
- Poormoghim H, Moradi Lakeh M, Mohammadipour M, Sodagari F, Toofaninjed N. Cyclophosphamide for scleroderma lung disease: a systematic review and meta-analysis. Rheumatology International 2012;32(8):2431-44. [PMID: ] - PubMed
Puyade 2019
-
- Puyade M, Maltez N, Lansiaux P, Pugnet G, Roblot P, Colmegna I. Health-related quality of life in systemic sclerosis before and after autologous haematopoietic stem cell transplant – a systematic review. Rheumatology 2019;59(4):779-89. - PubMed
Review Manager 2014 [Computer program]
-
- Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Schünemann 2021a
-
- Schünemann HJ, Vist GE, Higgins JP, Santesso N, Deeks JJ, Glasziou P, et al. Chapter 15: Interpreting results and drawing conclusions. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions. Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Schünemann 2021b
-
- Schünemann HJ, Higgins JP, Vist GE, Glasziou P, Akl EA, Skoetz N, et al. Chapter 14: Completing 'Summary of findings' tables and grading the certainty of the evidence. In Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Sekhon 2010
-
- Sekhon S, Pope J, Canadian Scleroderma Research Group, Baron M. The minimally important difference in clinical practice for patient-centered outcomes including Health Assessment Questionnaire, fatigue, pain, sleep, global visual analog scale, and SF-36 in scleroderma. Journal of Rheumatology 2010;37(3):591-8. - PubMed
Shouval 2018
-
- Shouval R, Furie N, Raanani P, Nagler A, Gafter-Gvili A. Autologous hematopoietic stem cell transplantation for systemic sclerosis: a systematic review and meta-analysis. Biology of Blood and Marrow Transplantation 2018;24(5):937-44. - PubMed
Snowden 2012
-
- Snowden JA, Saccardi R, Allez M, Ardizzone S, Arnold R, Cervera R, et al, EBMT Autoimmune Disease Working Party (ADWP), Paediatric Diseases Working Party (PDWP). Haematopoietic SCT in severe autoimmune diseases: updated guidelines of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplantation 2012;47(6):770-90. [PMID: ] - PMC - PubMed
Spierings 2022
Steen 1998
-
- Steen VD, Medsger TA Jr. Case-control study of corticosteroids and other drugs that either precipitate or protect from the development of scleroderma renal crisis. Arthritis and Rheumatism 1998;41(9):1613-9. [PMID: ] - PubMed
Steen 2000
-
- Steen VD, Medsger TA Jr. Severe organ involvement in systemic sclerosis with diffuse scleroderma. Arthritis and Rheumatism 2000;43(11):2437-44. [PMID: ] - PubMed
Tashkin 2006
-
- Tashkin DP, Elashoff R, Clements PJ, Goldin J, Roth MD, Furst DE, et al. Cyclophosphamide versus placebo in scleroderma lung disease. New England Journal of Medicine 2006;354(25):2655-66. - PubMed
Tashkin 2016
-
- Tashkin MP, Roth MD, Clements PJ, Khanna D, Kleerup EC, Golding K, et al. Mycophenolate mofetil versus oral cyclophosphamide in scleroderma-related interstitial lung disease (SLS II): a randomised controlled, double-blind, parallel group trial. Lancet Respiratory Medicine 2016;4(9):708-19. - PMC - PubMed
Toledano 2012
-
- Toledano E, Candelas G, Rosales Z, Martínez Prada C, León L, Abásolo L, et al. A meta-analysis of mortality in rheumatic diseases. Reumatologia Clinica 2012;8(6):334-41. [PMID: ] - PubMed
van den Hoogen 2013
van Laar 2013
-
- Laar JM, Sullivan K. Stem cell transplantation in systemic sclerosis. Current Opinion in Rheumatology 2013;25(6):719-25. [PMID: ] - PubMed
References to other published versions of this review
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

