The development of peripheral microvasculopathy with chronic metabolic disease in obese Zucker rats: a retrograde emergence?
- PMID: 35904886
- PMCID: PMC9448278
- DOI: 10.1152/ajpheart.00264.2022
The development of peripheral microvasculopathy with chronic metabolic disease in obese Zucker rats: a retrograde emergence?
Abstract
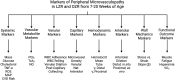
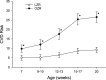
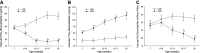
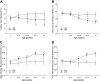
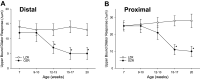
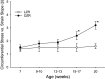
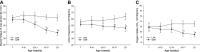
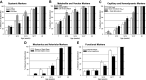
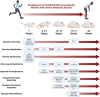
The study of peripheral vasculopathy with chronic metabolic disease is challenged by divergent contributions from spatial (the level of resolution or specific tissue being studied) and temporal origins (evolution of the developing impairments in time). Over many years of studying the development of skeletal muscle vasculopathy and its functional implications, we may be at the point of presenting an integrated conceptual model that addresses these challenges within the obese Zucker rat (OZR) model. At the early stages of metabolic disease, where systemic markers of elevated cardiovascular disease risk are present, the only evidence of vascular dysfunction is at postcapillary and collecting venules, where leukocyte adhesion/rolling is elevated with impaired venular endothelial function. As metabolic disease severity and duration increases, reduced microvessel density becomes evident as well as increased variability in microvascular hematocrit. Subsequently, hemodynamic impairments to distal arteriolar networks emerge, manifesting as increasing perfusion heterogeneity and impaired arteriolar reactivity. This retrograde "wave of dysfunction" continues, creating a condition wherein deficiencies to the distal arteriolar, capillary, and venular microcirculation stabilize and impairments to proximal arteriolar reactivity, wall mechanics, and perfusion distribution evolve. This proximal arteriolar dysfunction parallels increasing failure in fatigue resistance, hyperemic responses, and O2 uptake within self-perfused skeletal muscle. Taken together, these results present a conceptual model for the retrograde development of peripheral vasculopathy with chronic metabolic disease and provide insight into the timing and targeting of interventional strategies to improve health outcomes.NEW & NOTEWORTHY Working from an established database spanning multiple scales and times, we studied progression of peripheral microvascular dysfunction in chronic metabolic disease. The data implicate the postcapillary venular endothelium as the initiating site for vasculopathy. Indicators of dysfunction, spanning network structures, hemodynamics, vascular reactivity, and perfusion progress in an insidious retrograde manner to present as functional impairments to muscle blood flow and performance much later. The silent vasculopathy progression may provide insight into clinical treatment challenges.
Keywords: microcirculation; microvascular systems; peripheral vascular disease; rat models of metabolic syndrome; skeletal muscle fatigue.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures



References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

