Upregulation of the hypothalamo-neurohypophysial system and activation of vasopressin neurones attenuates hyperalgesia in a neuropathic pain model rat
- PMID: 35906406
- PMCID: PMC9338054
- DOI: 10.1038/s41598-022-17477-5
Upregulation of the hypothalamo-neurohypophysial system and activation of vasopressin neurones attenuates hyperalgesia in a neuropathic pain model rat
Abstract
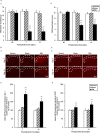
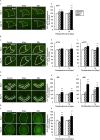
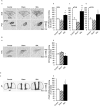
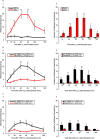
Arginine vasopressin (AVP) is a hypothalamic neurosecretory hormone well known as an antidiuretic, and recently reported to be involved in pain modulation. The expression kinetics of AVP and its potential involvement in the descending pain modulation system (DPMS) in neuropathic pain (NP) remains unclear. We investigated AVP expression and its effects on mechanical and thermal nociceptive thresholds using a unilateral spinal nerve ligation (SNL) model. All rats with SNL developed NP. Intensities of enhanced green fluorescent protein (eGFP) in the supraoptic and paraventricular nuclei, median eminence, and posterior pituitary were significantly increased at 7 and 14 days post-SNL in AVP-eGFP rats. In situ hybridisation histochemistry revealed significantly increased AVP mRNA expression at 14 days post-SNL compared with the sham control group. The chemogenetic activation of AVP neurones significantly attenuated mechanical and thermal hyperalgesia with elevated plasma AVP concentration. These analgesic effects were suppressed by pre-administration with V1a receptor antagonist. AVP neurones increased the neuronal activity of serotonergic dorsal raphe, noradrenergic locus coeruleus, and inhibitory interneurones in the spinal dorsal horn. These results suggest that the hypothalamo-neurohypophysial system of AVP is upregulated in NP and activated endogenous AVP exerts analgesic effects via the V1a receptors. AVP neurones may activate the DPMS.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
The neurohypophysial oxytocin and arginine vasopressin system is activated in a knee osteoarthritis rat model.J Neuroendocrinol. 2020 Aug;32(8):e12892. doi: 10.1111/jne.12892. Epub 2020 Aug 6. J Neuroendocrinol. 2020. PMID: 32761684
-
Acute Mono-Arthritis Activates the Neurohypophysial System and Hypothalamo-Pituitary Adrenal Axis in Rats.Front Endocrinol (Lausanne). 2020 Feb 11;11:43. doi: 10.3389/fendo.2020.00043. eCollection 2020. Front Endocrinol (Lausanne). 2020. PMID: 32117068 Free PMC article.
-
Arginine vasopressin induces analgesic effects and inhibits pyramidal cells in the anterior cingulate cortex in spared nerve injured mice.Am J Physiol Endocrinol Metab. 2024 Dec 1;327(6):E700-E710. doi: 10.1152/ajpendo.00361.2024. Epub 2024 Oct 23. Am J Physiol Endocrinol Metab. 2024. PMID: 39441239
-
Specific expression of optically active reporter gene in arginine vasopressin-secreting neurosecretory cells in the hypothalamic-neurohypophyseal system.J Neuroendocrinol. 2008 Jun;20(6):660-4. doi: 10.1111/j.1365-2826.2008.01706.x. J Neuroendocrinol. 2008. PMID: 18601686 Review.
-
New aspects of firing pattern autocontrol in oxytocin and vasopressin neurones.Adv Exp Med Biol. 1998;449:153-62. doi: 10.1007/978-1-4615-4871-3_18. Adv Exp Med Biol. 1998. PMID: 10026798 Review.
Cited by
-
Simultaneous Occurrence of Hyponatremia and Hypokalemia in a Patient with Herpes Zoster: A Case Report with a Review of the Literature.Intern Med. 2025 Jan 15;64(2):261-266. doi: 10.2169/internalmedicine.3795-24. Epub 2024 May 30. Intern Med. 2025. PMID: 38811217 Free PMC article. Review.
-
Copeptin in Acute Myocardial Infarction: Is There a Role in the Era of High-Sensitivity Troponins?J Cardiovasc Dev Dis. 2025 Apr 9;12(4):144. doi: 10.3390/jcdd12040144. J Cardiovasc Dev Dis. 2025. PMID: 40278203 Free PMC article. Review.
References
-
- Cuzzo B, Padala SA, Lappin SL. StatPearls. StatPearls Publishing © 2021, StatPearls Publishing LLC; 2021.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

