Co-cultivation of filamentous microorganisms in the presence of aluminum oxide microparticles
- PMID: 35906994
- PMCID: PMC9418094
- DOI: 10.1007/s00253-022-12087-7
Co-cultivation of filamentous microorganisms in the presence of aluminum oxide microparticles
Abstract
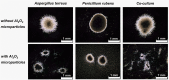
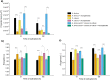
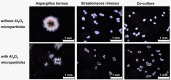
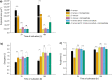
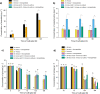
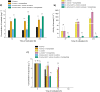
In the present work, the approaches of submerged co-cultivation and microparticle-enhanced cultivation (MPEC) were combined and evaluated over the course of three case studies. The filamentous fungus Aspergillus terreus was co-cultivated with Penicillium rubens, Streptomyces rimosus, or Cerrena unicolor in shake flasks with or without the addition of aluminum oxide microparticles. The influence of microparticles on the production of lovastatin, penicillin G, oxytetracycline, and laccase in co-cultures was compared with the effects recorded for the corresponding monocultures. In addition, the quantitative analyses of morphological parameters, sugars consumption, and by-products formation were performed. The study demonstrated that the influence of microparticles on the production of a given molecule in mono- and co-culture may differ considerably, e.g., the biosynthesis of oxytetracycline was shown to be inhibited due to the presence of aluminum oxide in "A. terreus vs. S. rimosus" co-cultivation variants but not in S. rimosus monocultures. The differences were also observed regarding the morphological characteristics, e.g., the microparticles-induced changes of projected area in the co-cultures and the corresponding monocultures were not always comparable. In addition, the study showed the importance of medium composition on the outcomes of MPEC, as exemplified by lovastatin production in A. terreus monocultures. Finally, the co-cultures of A. terreus with a white-rot fungus C. unicolor were described here for the first time. KEY POINTS: • Aluminum oxide affects secondary metabolites production in submerged co-cultures. • Mono- and co-cultures are differently impacted by the addition of aluminum oxide. • Effect of aluminum oxide on metabolites production depends on medium composition.
Keywords: Aspergillus terreus; Co-culture; Laccase; Lovastatin; Oxytetracycline; Penicillin.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Evaluating the outcomes of submerged co-cultivation: production of lovastatin and other secondary metabolites by Aspergillus terreus in fungal co-cultures.Appl Microbiol Biotechnol. 2019 Jul;103(14):5593-5605. doi: 10.1007/s00253-019-09874-0. Epub 2019 May 16. Appl Microbiol Biotechnol. 2019. PMID: 31098686 Free PMC article.
-
Enhanced Oxytetracycline Production by Streptomyces rimosus in Submerged Co-Cultures with Streptomyces noursei.Molecules. 2021 Oct 5;26(19):6036. doi: 10.3390/molecules26196036. Molecules. 2021. PMID: 34641580 Free PMC article.
-
Confrontation between Penicillium rubens and Aspergillus terreus: Investigating the production of fungal secondary metabolites in submerged co-cultures.J Biosci Bioeng. 2020 Nov;130(5):503-513. doi: 10.1016/j.jbiosc.2020.06.012. Epub 2020 Aug 2. J Biosci Bioeng. 2020. PMID: 32758403
-
Bioprocess Engineering Aspects of the Cultivation of a Lovastatin Producer Aspergillus terreus.Adv Biochem Eng Biotechnol. 2015;149:133-70. doi: 10.1007/10_2014_302. Adv Biochem Eng Biotechnol. 2015. PMID: 25633258 Review.
-
Modern morphological engineering techniques for improving productivity of filamentous fungi in submerged cultures.World J Microbiol Biotechnol. 2016 Dec;32(12):193. doi: 10.1007/s11274-016-2148-7. Epub 2016 Oct 7. World J Microbiol Biotechnol. 2016. PMID: 27718148 Free PMC article. Review.
Cited by
-
Analysis of secondary metabolites and morphology in Streptomyces rimosus microparticle-enhanced cultivation (MPEC) at various initial organic nitrogen concentrations.Microb Cell Fact. 2024 Sep 9;23(1):243. doi: 10.1186/s12934-024-02514-x. Microb Cell Fact. 2024. PMID: 39251992 Free PMC article.
-
Computation-aided studies related to the induction of specialized metabolite biosynthesis in microbial co-cultures: An introductory overview.Comput Struct Biotechnol J. 2023 Aug 16;21:4021-4029. doi: 10.1016/j.csbj.2023.08.011. eCollection 2023. Comput Struct Biotechnol J. 2023. PMID: 37649711 Free PMC article. Review.
References
-
- Askenazi M, Driggers EM, Holtzman DA, Norman TC, Iverson S, Zimmer DP, Boers ME, Blomquist PR, Martinez EJ, Monreal AW, Feibelman TP, Mayorga ME, Maxon ME, Sykes K, Tobin JV, Cordero E, Salama SR, Trueheart J, Royer JC, Madden KT. Integrating transcriptional and metabolite profiles to direct the engineering of lovastatin-producing fungal strains. Nat Biotechnol. 2003;21:150–156. doi: 10.1038/nbt781. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

