Molecular and biochemical regulation of skeletal muscle metabolism
- PMID: 35908794
- PMCID: PMC9339271
- DOI: 10.1093/jas/skac035
Molecular and biochemical regulation of skeletal muscle metabolism
Abstract
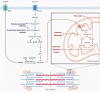
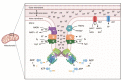
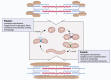
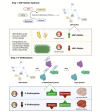
Skeletal muscle hypertrophy is a culmination of catabolic and anabolic processes that are interwoven into major metabolic pathways, and as such modulation of skeletal muscle metabolism may have implications on animal growth efficiency. Muscle is composed of a heterogeneous population of muscle fibers that can be classified by metabolism (oxidative or glycolytic) and contractile speed (slow or fast). Although slow fibers (type I) rely heavily on oxidative metabolism, presumably to fuel long or continuous bouts of work, fast fibers (type IIa, IIx, and IIb) vary in their metabolic capability and can range from having a high oxidative capacity to a high glycolytic capacity. The plasticity of muscle permits continuous adaptations to changing intrinsic and extrinsic stimuli that can shift the classification of muscle fibers, which has implications on fiber size, nutrient utilization, and protein turnover rate. The purpose of this paper is to summarize the major metabolic pathways in skeletal muscle and the associated regulatory pathways.
Keywords: metabolism; muscle; nutrients; satellite cells.
Plain language summary
Skeletal muscle is a heterogenous population of cells that are classified into muscle types based on contractile speed and metabolism. The various types of muscle cells utilize different biochemical pathways to produce energy to support cellular functions. These complex biochemical pathways are unique in their subcellular localization, substrate source, energy production capacity, and regulatory mechanisms. The purpose of this review is to describe the major metabolic pathways in skeletal muscle and the associated regulatory mechanisms.
© The Author(s) 2022. Published by Oxford University Press on behalf of the American Society of Animal Science. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures
Similar articles
-
Muscle mechanics: adaptations with exercise-training.Exerc Sport Sci Rev. 1996;24:427-73. Exerc Sport Sci Rev. 1996. PMID: 8744258 Review.
-
Distribution of fiber types determined by in situ hybridization of myosin heavy chain mRNA and enzyme histochemistry in rat skeletal muscles.Cell Mol Biol (Noisy-le-grand). 1997 May;43(3):319-27. Cell Mol Biol (Noisy-le-grand). 1997. PMID: 9193786
-
Response of the neuromuscular unit to spaceflight: what has been learned from the rat model.Exerc Sport Sci Rev. 1996;24:399-425. Exerc Sport Sci Rev. 1996. PMID: 8744257 Review.
-
Myosin heavy chain composition of different skeletal muscles in Large White and Meishan pigs.J Anim Sci. 2004 Jul;82(7):1931-41. doi: 10.2527/2004.8271931x. J Anim Sci. 2004. PMID: 15309939
-
Fiber composition and oxidative capacity of hamster skeletal muscle.J Histochem Cytochem. 2002 Dec;50(12):1685-92. doi: 10.1177/002215540205001214. J Histochem Cytochem. 2002. PMID: 12486092
Cited by
-
Exploring skeletal muscle tolerance and whole-body metabolic effects of FDA-approved drugs in a volumetric muscle loss model.Physiol Rep. 2023 Jun;11(12):e15756. doi: 10.14814/phy2.15756. Physiol Rep. 2023. PMID: 37332022 Free PMC article.
-
Possible Mechanisms Linking Obesity, Steroidogenesis, and Skeletal Muscle Dysfunction.Life (Basel). 2023 Jun 19;13(6):1415. doi: 10.3390/life13061415. Life (Basel). 2023. PMID: 37374197 Free PMC article. Review.
-
Development of an Alternative Protocol to Study Muscle Fatigue.Metabolites. 2025 Jan 16;15(1):54. doi: 10.3390/metabo15010054. Metabolites. 2025. PMID: 39852396 Free PMC article.
-
NC1184: Molecular Mechanisms Regulating Skeletal Muscle Growth and Differentiation.J Anim Sci. 2022 Aug 1;100(8):skac229. doi: 10.1093/jas/skac229. J Anim Sci. 2022. PMID: 35908784 Free PMC article. No abstract available.
-
The regulation of cell metabolism by hypoxia and hypercapnia.J Biol Chem. 2025 Mar;301(3):108252. doi: 10.1016/j.jbc.2025.108252. Epub 2025 Feb 4. J Biol Chem. 2025. PMID: 39914740 Free PMC article. Review.
References
-
- Acín-Pérez, R., Bayona-Bafaluy M. P., Fernández-Silva P., Moreno-Loshuertos R., Pérez-Martos A., Bruno C., Moraes C. T., and Enríquez J. A.. . 2004. Respiratory complex III is required to maintain complex I in mammalian mitochondria. Mol. Cell 13:805–815. doi:10.1016/s1097-2765(04)00124-8. - DOI - PMC - PubMed
-
- Amirouche, A., Durieux A. C., Banzet S., Koulmann N., Bonnefoy R., Mouret C., Bigard X., Peinnequin A., and Freyssenet D.. . 2009. Down-regulation of Akt/mammalian target of rapamycin signaling pathway in response to myostatin overexpression in skeletal muscle. Endocrinology 150:286–294. doi:10.1210/en.2008-0959. - DOI - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources

