Trichoderma stromaticum spores induce autophagy and downregulate inflammatory mediators in human peripheral blood-derived macrophages
- PMID: 35909603
- PMCID: PMC9325901
- DOI: 10.1016/j.crmicr.2022.100145
Trichoderma stromaticum spores induce autophagy and downregulate inflammatory mediators in human peripheral blood-derived macrophages
Abstract
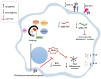
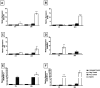
Trichoderma spp. are usually considered safe and normally used as biocontrol and biofertilization. Safety for human health is evaluated by several tests that detect various effects such as allergenicity, toxicity, infectivity, and pathogenicity. However, they do not evaluate the effects of the agent upon the immune system. The aim of this study was to investigate the interaction between T. stromaticum spores and mammalian cells to assess the immunomodulatory potential of the spores of this fungus. First, mouse macrophage cell line J774 and human macrophages were exposed to fungal spores and analyzed for structural features, through scanning and transmission electron microscopy. Then, various analysis were performed in human macrophages as to their effect in some functional and molecular aspects of the immune system through immunocytochemistry, flow cytometry and gene expression assays. We demonstrated that T. stromaticum spores induces autophagy and autophagy-related genes (ATGs) and downmodulate inflammatory mediators, including ROS, NLRP3, the cytokines IL-1β, IL-18, IL-12 and IL-10, as well as TLR2, TLR4, miR-146b and miR-155, which may lead to an augmented susceptibility to pathogens. Our study shows the extension of damages the biofungicide Tricovab® can cause in the innate immune response. Further studies are necessary to elucidate other innate and adaptive immune responses and, consequently, the safety of this fungus when in contact with humans.
Keywords: Autophagy, ROS, Inflammasomes; Biofungicide, Trichoderma; microRNAs.
© 2022 The Authors. Published by Elsevier B.V.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







References
-
- Alves-Filho E.R., Maioli T.U., Faria A.M.C., Noronha F.S.M., Silva N.M., Costa M.G.C., Santos J.L.Dos. The biocontrol fungus Trichoderma stromaticum downregulates respiratory burst and nitric oxide in phagocytes and IFN-gamma and IL-10. J. Toxicol. Environ. Health. A. 2011;74:943–958. doi: 10.1080/15287394.2011.573747. - DOI - PubMed
-
- Andersen C.L., Jensen J.L., Ørntoft T.F. Normalization of real-time quantitative reverse transcription-PCR data: A model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res. 2004;64:5245–5250. https://doi.org/10.1158/0008-5472.CAN-04-0496. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

