Enzymatic Production of Chondroitin Oligosaccharides and Its Sulfate Derivatives
- PMID: 35910011
- PMCID: PMC9326237
- DOI: 10.3389/fbioe.2022.951740
Enzymatic Production of Chondroitin Oligosaccharides and Its Sulfate Derivatives
Abstract
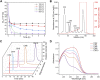
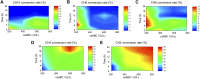
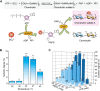
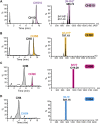
Chondroitin sulfate (CS) has a wide range of physiological functions and clinical applications. However, the biosynthesis of chondroitin oligosaccharides (o-CHs) and sulfate derivatives with specific length is always challenging. Herein, we report enzymatic strategies for producing homogeneous o-CHs and its sulfate derivatives from microbial sourced chondroitin. Chondroitin disaccharides, tetrasaccharides, hexasaccharides, octasaccharides, and decasaccharides with defined structure were produced by controllably depolymerizing microbial sourced chondroitin with an engineered chondroitinase ABC I. The highest conversion rates of the above corresponding o-CHs were 65.5%, 32.1%, 12.7%, 7.2%, and 16.3%, respectively. A new efficient enzymatic sulfation system that directly initiates from adenosine 5'-triphosphate (ATP) and sulfate was developed and improved the sulfation of chondroitin from 8.3% to 85.8% by optimizing the temperature, sulfate and ATP concentration. o-CHs decasaccharide, octasaccharide, hexasaccharide, tetrasaccharide and disaccharide were modified and the corresponding sulfate derivatives with one sulfate group were prepared. The enzymatic approaches constructed here for preparing o-CHs and its sulfate derivatives pave the way for the study of structure-activity relationship and applications.
Keywords: chondroitin; chondroitin sulfate; chondroitinase ABC I; oligosaccharides; sulfation system.
Copyright © 2022 Zhang, Xu, Jin, Wang, Hu, Zhang, Du and Kang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Characteristic hexasaccharide sequences in octasaccharides derived from shark cartilage chondroitin sulfate D with a neurite outgrowth promoting activity.J Biol Chem. 1998 Feb 6;273(6):3296-307. doi: 10.1074/jbc.273.6.3296. J Biol Chem. 1998. PMID: 9452446
-
Two related but distinct chondroitin sulfate mimetope octasaccharide sequences recognized by monoclonal antibody WF6.J Biol Chem. 2007 Nov 30;282(48):35232-46. doi: 10.1074/jbc.M702255200. Epub 2007 Sep 19. J Biol Chem. 2007. PMID: 17884822
-
Novel tetrasaccharides isolated from squid cartilage chondroitin sulfate E contain unusual sulfated disaccharide units GlcA(3-O-sulfate)beta1-3GalNAc(6-O-sulfate) or GlcA(3-O-sulfate)beta1-3GalNAc.J Biol Chem. 1997 Aug 8;272(32):19656-65. doi: 10.1074/jbc.272.32.19656. J Biol Chem. 1997. PMID: 9242620
-
Advances and challenges in biotechnological production of chondroitin sulfate and its oligosaccharides.Int J Biol Macromol. 2023 Dec 31;253(Pt 1):126551. doi: 10.1016/j.ijbiomac.2023.126551. Epub 2023 Sep 1. Int J Biol Macromol. 2023. PMID: 37659488 Review.
-
Biosynthesis and function of chondroitin sulfate.Biochim Biophys Acta. 2013 Oct;1830(10):4719-33. doi: 10.1016/j.bbagen.2013.06.006. Epub 2013 Jun 14. Biochim Biophys Acta. 2013. PMID: 23774590 Review.
Cited by
-
Engineering sulfonate group donor regeneration systems to boost biosynthesis of sulfated compounds.Nat Commun. 2023 Nov 10;14(1):7297. doi: 10.1038/s41467-023-43195-1. Nat Commun. 2023. PMID: 37949843 Free PMC article.
-
Efficient Expression and Characterization of an Endo-Type Lyase HCLase_M28 and Its Gradual Scale-Up Fermentation for the Preparation of Chondroitin Sulfate Oligosaccharides.Appl Biochem Biotechnol. 2024 Sep;196(9):6526-6555. doi: 10.1007/s12010-024-04878-7. Epub 2024 Feb 22. Appl Biochem Biotechnol. 2024. PMID: 38386140
-
Biomimetic Proteoglycans for Intervertebral Disc (IVD) Regeneration.Biomimetics (Basel). 2024 Nov 22;9(12):722. doi: 10.3390/biomimetics9120722. Biomimetics (Basel). 2024. PMID: 39727726 Free PMC article. Review.
-
Enhanced hydrogel loading of quercetin-loaded hollow mesoporous cerium dioxide nanoparticles for skin flap survival.Mater Today Bio. 2024 Dec 28;30:101432. doi: 10.1016/j.mtbio.2024.101432. eCollection 2025 Feb. Mater Today Bio. 2024. PMID: 39839491 Free PMC article.
-
Chondroitin Sulfate Nanovectorized by LC-PUFAs Nanocarriers Extracted from Salmon (Salmo salar) by Green Process with Decreased Inflammatory Marker Expression in Interleukin-1β-Stimulated Primary Human Chondrocytes In Vitro Culture.Mar Drugs. 2024 Dec 20;22(12):571. doi: 10.3390/md22120571. Mar Drugs. 2024. PMID: 39728145 Free PMC article.
References
-
- Cimini D., Iacono I. D., Carlino E., Finamore R., Restaino O. F., Diana P., et al. (2017). Engineering S. Equi Subsp. Zooepidemicus towards Concurrent Production of Hyaluronic Acid and Chondroitin Biopolymers of Biomedical Interest. Amb. Expr. 7 (1), 61. 10.1186/s13568-017-0364-7 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

