Capsule type defines the capability of Klebsiella pneumoniae in evading Kupffer cell capture in the liver
- PMID: 35914009
- PMCID: PMC9342791
- DOI: 10.1371/journal.ppat.1010693
Capsule type defines the capability of Klebsiella pneumoniae in evading Kupffer cell capture in the liver
Abstract
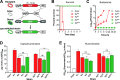
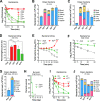
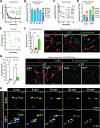
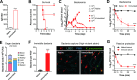
Polysaccharide capsule is the main virulence factor of K. pneumoniae, a major pathogen of bloodstream infections in humans. While more than 80 capsular serotypes have been identified in K. pneumoniae, only several serotypes are frequently identified in invasive infections. It is documented that the capsule enhances bacterial resistance to phagocytosis, antimicrobial peptides and complement deposition under in vitro conditions. However, the precise role of the capsule in the process of K. pneumoniae bloodstream infections remains to be elucidated. Here we show that the capsule promotes K. pneumoniae survival in the bloodstream by protecting bacteria from being captured by liver resident macrophage Kupffer cells (KCs). Our real-time in vivo imaging revealed that blood-borne acapsular K. pneumoniae mutant is rapidly captured and killed by KCs in the liver sinusoids of mice, whereas, to various extents, encapsulated strains bypass the anti-bacterial machinery in a serotype-dependent manner. Using capsule switched strains, we show that certain high-virulence (HV) capsular serotypes completely block KC's capture, whereas the low-virulence (LV) counterparts confer partial protection against KC's capture. Moreover, KC's capture of the LV K. pneumoniae could be in vivo neutralized by free capsular polysaccharides of homologous but not heterologous serotypes, indicating that KCs specifically recognize the LV capsules. Finally, immunization with inactivated K. pneumoniae enables KCs to capture the HV K. pneumoniae. Together, our findings have uncovered that KCs are the major target cells of K. pneumoniae capsule to promote bacterial survival and virulence, which can be reversed by vaccination.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




Similar articles
-
Functional vulnerability of liver macrophages to capsules defines virulence of blood-borne bacteria.J Exp Med. 2022 Apr 4;219(4):e20212032. doi: 10.1084/jem.20212032. Epub 2022 Mar 8. J Exp Med. 2022. PMID: 35258552 Free PMC article.
-
Natural antibodies to polysaccharide capsules enable Kupffer cells to capture invading bacteria in the liver sinusoids.J Exp Med. 2025 Feb 3;222(2):e20240735. doi: 10.1084/jem.20240735. Epub 2024 Dec 24. J Exp Med. 2025. PMID: 39718543 Free PMC article.
-
Capsular polysaccharide enables Klebsiella pneumoniae to evade phagocytosis by blocking host-bacteria interactions.mBio. 2025 Mar 12;16(3):e0383824. doi: 10.1128/mbio.03838-24. Epub 2025 Feb 14. mBio. 2025. PMID: 39950808 Free PMC article.
-
[Capsular polysaccharide of Klebsiella pneumoniae. II. Immunogenic properties].Rev Latinoam Microbiol. 1993 Jan-Mar;35(1):109-15. Rev Latinoam Microbiol. 1993. PMID: 8140330 Review. Spanish.
-
Molecular pathogenesis of Klebsiella pneumoniae.Future Microbiol. 2014;9(9):1071-81. doi: 10.2217/fmb.14.48. Future Microbiol. 2014. PMID: 25340836 Review.
Cited by
-
Therapeutic efficacy of a K5-specific phage and depolymerase against Klebsiella pneumoniae in a mouse model of infection.Vet Res. 2024 May 7;55(1):59. doi: 10.1186/s13567-024-01311-z. Vet Res. 2024. PMID: 38715095 Free PMC article.
-
Carbapenem-Resistant Klebsiella pneumoniae: Virulence Factors, Molecular Epidemiology and Latest Updates in Treatment Options.Antibiotics (Basel). 2023 Jan 21;12(2):234. doi: 10.3390/antibiotics12020234. Antibiotics (Basel). 2023. PMID: 36830145 Free PMC article. Review.
-
Klebsiella pneumoniae causes bacteremia using factors that mediate tissue-specific fitness and resistance to oxidative stress.PLoS Pathog. 2023 Jul 18;19(7):e1011233. doi: 10.1371/journal.ppat.1011233. eCollection 2023 Jul. PLoS Pathog. 2023. PMID: 37463183 Free PMC article.
-
Genome-wide phage susceptibility analysis in Acinetobacter baumannii reveals capsule modulation strategies that determine phage infectivity.PLoS Pathog. 2023 Jun 8;19(6):e1010928. doi: 10.1371/journal.ppat.1010928. eCollection 2023 Jun. PLoS Pathog. 2023. PMID: 37289824 Free PMC article.
-
Poor Glycemic Control in Carbapenem-Resistant Klebsiella pneumoniae Infections: Impact on Epidemiological Features, Mortality Risks, and Polymyxin Resistance.Infect Drug Resist. 2025 Feb 1;18:647-660. doi: 10.2147/IDR.S501632. eCollection 2025. Infect Drug Resist. 2025. PMID: 39916694 Free PMC article.
References
-
- Russo TA, Olson R, Fang CT, Stoesser N, Miller M, MacDonald U, et al.. Identification of biomarkers for differentiation of hypervirulent Klebsiella pneumoniae from Classical K. pneumoniae. J Clin Microbiol. 2018;56(9):e00776–18. Epub 2018/06/22. doi: 10.1128/JCM.00776-18 ; PubMed Central PMCID: PMC6113484. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

