P38α MAPK is a gatekeeper of uterine progesterone responsiveness at peri-implantation via Ube3c-mediated PGR degradation
- PMID: 35914132
- PMCID: PMC9371708
- DOI: 10.1073/pnas.2206000119
P38α MAPK is a gatekeeper of uterine progesterone responsiveness at peri-implantation via Ube3c-mediated PGR degradation
Abstract
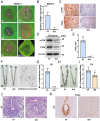
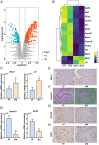
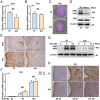
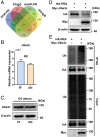
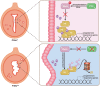
Estrogen and progesterone specify the establishment of uterine receptivity mainly through their respective nuclear receptors, ER and PR. PR is transcriptionally induced by estrogen-ER signaling in the endometrium, but how the protein homeostasis of PR in the endometrium is regulated remains elusive. Here, we demonstrated that the uterine-selective depletion of P38α derails normal uterine receptivity ascribed to the dramatic down-regulation of PR protein and disordered progesterone responsiveness in the uterine stromal compartment, leading to defective implantation and female infertility. Specifically, Ube3c, an HECT family E3 ubiquitin ligase, targets PR for polyubiquitination and thus proteasome degradation in the absence of P38α. Moreover, we discovered that P38α restrains the polyubiquitination activity of Ube3c toward PR by phosphorylating the Ube3c at serine741 . In summary, we provided genetic evidence for the regulation of PR protein stability in the endometrium by P38α and identified Ube3c, whose activity was modulated by P38α-mediated phosphorylation, as an E3 ubiquitin ligase for PR in the uterus.
Keywords: P38α; Ube3c; progesterone receptor; uterine receptivity.
Conflict of interest statement
The authors declare no competing interest.
Figures



Similar articles
-
Uterine Epithelial Progesterone Receptor Governs Uterine Receptivity Through Epithelial Cell Differentiation.Endocrinology. 2020 Dec 1;161(12):bqaa195. doi: 10.1210/endocr/bqaa195. Endocrinology. 2020. PMID: 33099617
-
Polycomb subunit BMI1 determines uterine progesterone responsiveness essential for normal embryo implantation.J Clin Invest. 2018 Jan 2;128(1):175-189. doi: 10.1172/JCI92862. Epub 2017 Nov 20. J Clin Invest. 2018. PMID: 29202468 Free PMC article.
-
WT1 directs normal progesterone receptor-chromatin binding essential for uterine receptivity at peri-implantation.Proc Natl Acad Sci U S A. 2025 Jul 15;122(28):e2504361122. doi: 10.1073/pnas.2504361122. Epub 2025 Jul 8. Proc Natl Acad Sci U S A. 2025. PMID: 40627402
-
Role of nuclear progesterone receptor isoforms in uterine pathophysiology.Hum Reprod Update. 2015 Mar-Apr;21(2):155-73. doi: 10.1093/humupd/dmu056. Epub 2014 Nov 18. Hum Reprod Update. 2015. PMID: 25406186 Free PMC article. Review.
-
Implantation in the baboon: endometrial responses.Semin Reprod Endocrinol. 1999;17(3):257-65. doi: 10.1055/s-2007-1016233. Semin Reprod Endocrinol. 1999. PMID: 10797944 Review.
Cited by
-
Phosphorylation of IDH1 Facilitates Progestin Resistance in Endometrial Cancer.Adv Sci (Weinh). 2024 Jun;11(23):e2310208. doi: 10.1002/advs.202310208. Epub 2024 Apr 6. Adv Sci (Weinh). 2024. PMID: 38582508 Free PMC article.
-
Targeting TTK Inhibits Tumorigenesis of T-Cell Lymphoma Through Dephosphorylating p38α and Activating AMPK/mTOR Pathway.Adv Sci (Weinh). 2025 Mar;12(10):e2413990. doi: 10.1002/advs.202413990. Epub 2025 Jan 21. Adv Sci (Weinh). 2025. PMID: 39836493 Free PMC article.
-
Varied cellular abnormalities in thin vs. normal endometrium in recurrent implantation failure by single-cell transcriptomics.Reprod Biol Endocrinol. 2024 Jul 31;22(1):90. doi: 10.1186/s12958-024-01263-1. Reprod Biol Endocrinol. 2024. PMID: 39085925 Free PMC article.
-
Stromal Pbrm1 mediates chromatin remodeling necessary for embryo implantation in the mouse uterus.J Clin Invest. 2024 Mar 1;134(5):e174194. doi: 10.1172/JCI174194. J Clin Invest. 2024. PMID: 38426493 Free PMC article.
-
Basic Research Advances in China on Embryo Implantation, Placentation, and Parturition.Matern Fetal Med. 2024 Jan 15;6(1):37-49. doi: 10.1097/FM9.0000000000000210. eCollection 2024 Jan. Matern Fetal Med. 2024. PMID: 40406740 Free PMC article.
References
-
- Wang H., Dey S. K., Roadmap to embryo implantation: Clues from mouse models. Nat. Rev. Genet. 7, 185–199 (2006). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

