Preclinical and Clinical Amelioration of Bone Fractures with Mesenchymal Stromal Cells: a Systematic Review and Meta-Analysis
- PMID: 35916286
- PMCID: PMC9350497
- DOI: 10.1177/09636897211051743
Preclinical and Clinical Amelioration of Bone Fractures with Mesenchymal Stromal Cells: a Systematic Review and Meta-Analysis
Abstract
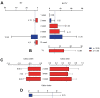
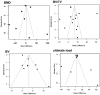
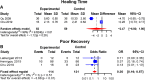
Even though reunion of bone fracture confronts clinicians, mesenchymal stromal cells (MSCs) are investigated to be curative in bone fracture. This study aimed to explore the application potential of MSCs for healing bone fractures. By inputting search terms and retrieving studies published up to March 2021, multiple databases, including PubMed, EMBASE, Web of Science, and Cochrane Library, were searched to identify eligible studies. The mean difference (MD) and 95% confidence interval (95% CI) were calculated to analyze the main results in the meta-analysis. Data analysis was performed using Engauge Digitizer 10.8 and R Software. Of the 31 articles, 26 were preclinical studies (n = 913), and 5 were clinical trials (n = 335). Preclinically, MSCs therapy significantly augmented the progress of bone regeneration [(bone volume over tissue volume (MD7.35, p < 0.01)], despite some non-significant effects (on the callus index, bone strength, work to failure, and stiffness). Clinically, the MSC group had a significantly reduced incidence of poor recovery (odds ratio (OR) 0.30, p < 0.01); however, a significant decrease in healing time was not observed in the MSC group (MD 2.47, p = 0.26). In summary, our data suggest that patients with bone fractures benefited from MSC administration and that MSCs are a potentially useful agent for bone regeneration. Despite these satisfactory outcomes, larger randomised clinical trials (RCTs) are necessary to confirm these findings.
Keywords: bone regeneration; clinical trials; mesenchymal stromal cells; meta-analysis; preclinical trials.
Conflict of interest statement
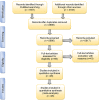
Figures

Similar articles
-
Clinical studies investigating the role of mesenchymal stem cells in healing of fracture non-unions: a systematic review.J Pak Med Assoc. 2023 Feb;73(Suppl 1)(2):S26-S31. doi: 10.47391/JPMA.AKUS-05. J Pak Med Assoc. 2023. PMID: 36788388
-
A Systematic Review and Meta-Analysis: Safety and Efficacy of Mesenchymal Stem Cells Therapy for Heart Failure.Curr Stem Cell Res Ther. 2021;16(3):354-365. doi: 10.2174/1574888X15999200820171432. Curr Stem Cell Res Ther. 2021. PMID: 32867655
-
Stem Cells and Their Derivatives: An Implication for the Regeneration of Nonunion Fractures.Cell Transplant. 2023 Jan-Dec;32:9636897231183530. doi: 10.1177/09636897231183530. Cell Transplant. 2023. PMID: 37462248 Free PMC article. Review.
-
Intra-Articular Mesenchymal Stromal Cell Injections Are No Different From Placebo in the Treatment of Knee Osteoarthritis: A Systematic Review and Meta-analysis of Randomized Controlled Trials.Arthroscopy. 2021 Jan;37(1):340-358. doi: 10.1016/j.arthro.2020.10.016. Epub 2020 Oct 21. Arthroscopy. 2021. PMID: 33098949
-
Role of mesenchymal stem cells in bone regeneration and fracture repair: a review.Int Orthop. 2013 Dec;37(12):2491-8. doi: 10.1007/s00264-013-2059-2. Epub 2013 Aug 15. Int Orthop. 2013. PMID: 23948983 Free PMC article. Review.
Cited by
-
Immunomodulation Using BMP-7 and IL-10 to Enhance the Mineralization Capacity of Bone Progenitor Cells in a Fracture Hematoma-Like Environment.Adv Healthc Mater. 2025 Feb;14(5):e2400077. doi: 10.1002/adhm.202400077. Epub 2024 Apr 15. Adv Healthc Mater. 2025. PMID: 38599586 Free PMC article.
-
Preclinical Assessment in Juvenile Sheep of an Allogeneic Bone Tissue Engineering Product with Wharton's Jelly Mesenchymal Stromal Cells.Cells. 2025 Jun 7;14(12):862. doi: 10.3390/cells14120862. Cells. 2025. PMID: 40558490 Free PMC article.
-
The Use of MSCs, iPSCs, and EVs in the Repair of Human MSK Tissues: Is Ultimate Success Dependent on Developing Excellent Implant Materials as Well as Creating an Optimal Environment for Implantation? What Is the Rationale for These Choices?Int J Mol Sci. 2025 Jun 28;26(13):6250. doi: 10.3390/ijms26136250. Int J Mol Sci. 2025. PMID: 40650028 Free PMC article. Review.
-
Human Wharton's jelly-derived mesenchymal stromal cells promote bone formation in immunodeficient mice when administered into a bone microenvironment.J Transl Med. 2023 Nov 10;21(1):802. doi: 10.1186/s12967-023-04672-9. J Transl Med. 2023. PMID: 37950242 Free PMC article.
-
Methods to accelerate fracture healing - a narrative review from a clinical perspective.Front Immunol. 2024 Jun 7;15:1384783. doi: 10.3389/fimmu.2024.1384783. eCollection 2024. Front Immunol. 2024. PMID: 38911851 Free PMC article. Review.
References
-
- Court-Brown CM, McQueen MM. Global forum: fractures in the elderly. J Bone Joint Surg Am. 2016;98(9):e36. - PubMed
-
- Griffin XL, Costa ML, Parsons N, Smith N. Electromagnetic field stimulation for treating delayed union or non-union of long bone fractures in adults. Cochrane Database Syst Rev. 2011;(4):D8471. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

