Induction of Interferon-Stimulated Genes Correlates with Reduced Growth of Influenza A Virus in Lungs after RIG-I Agonist Treatment of Ferrets
- PMID: 35916513
- PMCID: PMC9400473
- DOI: 10.1128/jvi.00559-22
Induction of Interferon-Stimulated Genes Correlates with Reduced Growth of Influenza A Virus in Lungs after RIG-I Agonist Treatment of Ferrets
Abstract
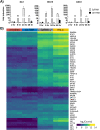
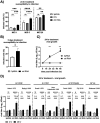
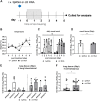
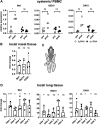
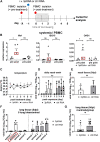
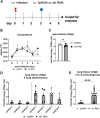
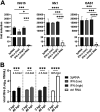
Intracellular RIG-I receptors represent key innate sensors of RNA virus infection, and RIG-I activation results in the induction of hundreds of host effector genes, including interferon-stimulated genes (ISGs). Synthetic RNA agonists targeting RIG-I have shown promise as antivirals against a broad spectrum of viruses, including influenza A virus (IAV), in both in vitro and mouse models of infection. Herein, we demonstrate that treatment of a ferret airway epithelial (FRL) cell line with a RIG-I agonist rapidly and potently induced expression of a broad range of ISGs and resulted in potent inhibition of growth of different IAV strains. In ferrets, a single intravenous injection of RIG-I agonist was associated with upregulated ISG expression in peripheral blood mononuclear cells and lung tissue, but not in nasal tissues. In a ferret model of viral contact transmission, a single treatment of recipient animals 24 h prior to cohousing with IAV-infected donors did not reduce virus transmission and shedding but did result in reduced lung virus titers 6 days after treatment. A single treatment of the IAV-infected donor animals also resulted in reduced virus titers in the lungs 2 days later. Thus, a single intravenous treatment with RIG-I agonist prior to infection or to ferrets with an established IAV infection can reduce virus growth in the lungs. These findings support further development of RIG-I agonists as effective antiviral treatments to limit the impact of IAV infections, particularly in reducing virus replication in the lower airways. IMPORTANCE RIG-I agonists have shown potential as broad-spectrum antivirals in vitro and in mouse models of infection. However, their antiviral potential has not been reported in outbred animals such as ferrets, which are widely regarded as the gold standard small animal model for human IAV infections. Herein, we demonstrate that RIG-I agonist treatment of a ferret airway cell line resulted in ISG induction and inhibition of a broad range of human influenza viruses. A single intravenous treatment of ferrets also resulted in systemic induction of ISGs, including in lung tissue, and when delivered to animals prior to IAV exposure or to animals with established IAV infection treatment resulted in reduced virus replication in the lungs. These data demonstrate the effectiveness of single RIG-I treatment against IAV in the ferret model and highlight the importance of future studies to optimize treatment regimens and delivery routes to maximize their ability to ameliorate IAV infections.
Keywords: RIG-I; antiviral agents; ferret; influenza; innate immunity; mouse model.
Conflict of interest statement
The authors declare a conflict of interest. G.H. holds patents on RIG-I and is one of the founders of the Rigontec GmbH.
Figures
Similar articles
-
Induction and antiviral activity of ferret myxovirus resistance (Mx) protein 1 against influenza A viruses.Sci Rep. 2024 Jun 12;14(1):13524. doi: 10.1038/s41598-024-63314-2. Sci Rep. 2024. PMID: 38866913 Free PMC article.
-
Expression of a Functional Mx1 Protein Is Essential for the Ability of RIG-I Agonist Prophylaxis to Provide Potent and Long-Lasting Protection in a Mouse Model of Influenza A Virus Infection.Viruses. 2022 Jul 15;14(7):1547. doi: 10.3390/v14071547. Viruses. 2022. PMID: 35891527 Free PMC article.
-
Delay of innate immune responses following influenza B virus infection affects the development of a robust antibody response in ferrets.mBio. 2025 Feb 5;16(2):e0236124. doi: 10.1128/mbio.02361-24. Epub 2025 Jan 8. mBio. 2025. PMID: 39772665 Free PMC article.
-
Modulation of Innate Immune Responses by the Influenza A NS1 and PA-X Proteins.Viruses. 2018 Dec 12;10(12):708. doi: 10.3390/v10120708. Viruses. 2018. PMID: 30545063 Free PMC article. Review.
-
Host Innate Antiviral Response to Influenza A Virus Infection: From Viral Sensing to Antagonism and Escape.Pathogens. 2024 Jul 3;13(7):561. doi: 10.3390/pathogens13070561. Pathogens. 2024. PMID: 39057788 Free PMC article. Review.
Cited by
-
Induction and antiviral activity of ferret myxovirus resistance (Mx) protein 1 against influenza A viruses.Sci Rep. 2024 Jun 12;14(1):13524. doi: 10.1038/s41598-024-63314-2. Sci Rep. 2024. PMID: 38866913 Free PMC article.
-
Immunoengineering of a Photocaged 5´-triphosphate Oligoribonucleotide Ligand for Spatiotemporal Control of RIG-I Activation in Cancer.Angew Chem Int Ed Engl. 2025 May;64(21):e202423321. doi: 10.1002/anie.202423321. Epub 2025 Apr 21. Angew Chem Int Ed Engl. 2025. PMID: 40095771 Free PMC article.
References
-
- Schlee M, Roth A, Hornung V, Hagmann CA, Wimmenauer V, Barchet W, Coch C, Janke M, Mihailovic A, Wardle G, Juranek S, Kato H, Kawai T, Poeck H, Fitzgerald KA, Takeuchi O, Akira S, Tuschl T, Latz E, Ludwig J, Hartmann G. 2009. Recognition of 5' triphosphate by RIG-I helicase requires short blunt double-stranded RNA as contained in panhandle of negative-strand virus. Immunity 31:25–34. 10.1016/j.immuni.2009.05.008. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

