Recent advances in factor XII structure and function
- PMID: 35916558
- PMCID: PMC9350914
- DOI: 10.1097/MOH.0000000000000727
Recent advances in factor XII structure and function
Abstract
Purpose of review: Factor XII (FXII), the precursor of the protease FXIIa, contributes to pathologic processes including angioedema and thrombosis. Here, we review recent work on structure-function relationships for FXII based on studies using recombinant FXII variants.
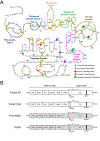
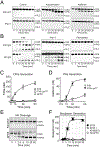
Recent findings: FXII is a homolog of pro-hepatocyte growth factor activator (Pro-HGFA). We prepared FXII in which domains are replaced by corresponding parts of Pro-HGA, and tested them in FXII activation and activity assays. In solution, FXII and prekallikrein undergo reciprocal activation to FXIIa and kallikrein. The rate of this process is restricted by the FXII fibronectin type-2 and kringle domains. Pro-HGA replacements for these domains accelerate FXII and prekallikrein activation. When FXII and prekallikrein bind to negatively charged surfaces, reciprocal activation is enhanced. The FXII EGF1 domain is required for surface binding.
Summary: We propose a model in which FXII is normally maintained in a closed conformation resistant to activation by intramolecular interactions involving the fibronectin type-2 and kringle domains. These interactions are disrupted when FXII binds to a surface through EGF1, enhancing FXII activation and prekallikrein activation by FXIIa. These observations have important implications for understanding the contributions of FXII to disease, and for developing therapies to treat thrombo-inflammatory disorders.
Copyright © 2022 Wolters Kluwer Health, Inc. All rights reserved.
Conflict of interest statement
CONFLICTS OF INTEREST
A.S. and M.L have no conflicts to report. D.G. is a consultant for several pharmaceutical companies that are developing compounds that target factor XI and factor XII for therapeutic purposes.
Figures



References
-
-
Maas C, Renne T. Coagulation factor XII in thrombosis and inflammation. Blood. 2018;131(17):1903–1909.
✶An excellent summary on the contributions of factor XII and contact activation to thombo-inflammatory disorders.
-
-
-
Shamanaev A, Ivanov I, Sun MF, et al. A Model for Surface-Dependent Factor XII Activation: The Roles of Factor XII Heavy Chain Domains. Blood Adv. (in press).
✶This manuscript contains details on most of the structure-function data for FXII covered in this review.
-
-
- Gailani D, Wheeler AP, Neff AT. Rare coagulation factor deficiencies. In: Hoffman R, Benz EJ, Silberstein LE, et al. , eds. Hematology: Basic Principles and Practice. 7th ed. Philadelphia, PA: Elsevier; 2018: 2034–2050
-
- Schmaier AH. The contact activation and kallikrein/kinin systems: pathophysiologic and physiologic activities. J Thromb Haemost. 2016;14(1):28–39. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

