Synthetic PPAR Agonist DTMB Alleviates Alzheimer's Disease Pathology by Inhibition of Chronic Microglial Inflammation in 5xFAD Mice
- PMID: 35917087
- PMCID: PMC9606171
- DOI: 10.1007/s13311-022-01275-y
Synthetic PPAR Agonist DTMB Alleviates Alzheimer's Disease Pathology by Inhibition of Chronic Microglial Inflammation in 5xFAD Mice
Abstract
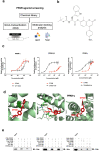
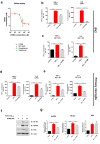
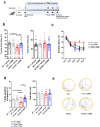
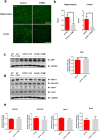
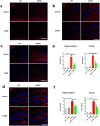
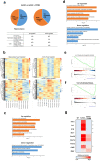
Abnormal productions of amyloid beta (Aβ) plaque and chronic neuroinflammation are commonly observed in the brain of patients with Alzheimer's disease, and both of which induce neuronal cell death, loss of memory, and cognitive dysfunction. However, many of the drugs targeting the production of Aβ peptides have been unsuccessful in treating Alzheimer's disease. In this study, we identified synthetic novel peroxisome proliferator-activating receptor (PPAR) agonist, DTMB, which can ameliorate the chronic inflammation and Aβ pathological progression of Alzheimer's disease. We discovered that DTMB attenuated the proinflammatory cytokine production of microglia by reducing the protein level of NF-κB. DTMB also improved the learning and memory defects and reduced the amount of Aβ plaque in the brain of 5xFAD mice. This reduction in Aβ pathology was attributed to the changes in gliosis and chronic inflammation level. Additionally, bulk RNA-sequencing showed that genes related to inflammation and cognitive function were changed in the hippocampus and cortex of DTMB-treated mice. Our findings demonstrate that DTMB has the potential to be a novel therapeutic agent for Alzheimer's disease.
Keywords: Alzheimer's disease; Astrocyte; Microglia; Neuroinflammation; PPAR.
© 2022. The Author(s).
Figures


Similar articles
-
The neuroprotective N-terminal amyloid-β core hexapeptide reverses reactive gliosis and gliotoxicity in Alzheimer's disease pathology models.J Neuroinflammation. 2023 May 27;20(1):129. doi: 10.1186/s12974-023-02807-9. J Neuroinflammation. 2023. PMID: 37245024 Free PMC article.
-
Activation of the nuclear receptor PPARδ is neuroprotective in a transgenic mouse model of Alzheimer's disease through inhibition of inflammation.J Neuroinflammation. 2015 Jan 16;12:7. doi: 10.1186/s12974-014-0229-9. J Neuroinflammation. 2015. PMID: 25592770 Free PMC article.
-
Fibrillar Aβ triggers microglial proteome alterations and dysfunction in Alzheimer mouse models.Elife. 2020 Jun 8;9:e54083. doi: 10.7554/eLife.54083. Elife. 2020. PMID: 32510331 Free PMC article.
-
Effects of CX3CR1 and Fractalkine Chemokines in Amyloid Beta Clearance and p-Tau Accumulation in Alzheimer's Disease (AD) Rodent Models: Is Fractalkine a Systemic Biomarker for AD?Curr Alzheimer Res. 2016;13(4):403-12. doi: 10.2174/1567205013666151116125714. Curr Alzheimer Res. 2016. PMID: 26567742 Review.
-
PPARs (Peroxisome Proliferator-activated Receptors) and Their Agonists in Alzheimer's Disease.Med Chem. 2024;20(8):781-798. doi: 10.2174/0115734064295063240422100615. Med Chem. 2024. PMID: 38726789 Review.
Cited by
-
Oleoylethanolamide facilitates PPARa and TFEB signaling and attenuates Ab pathology in a mouse model of Alzheimer's disease.Res Sq [Preprint]. 2023 Jan 20:rs.3.rs-2484513. doi: 10.21203/rs.3.rs-2484513/v1. Res Sq. 2023. Update in: Mol Neurodegener. 2023 Aug 15;18(1):56. doi: 10.1186/s13024-023-00648-x. PMID: 36711875 Free PMC article. Updated. Preprint.
-
Pathogenesis and therapeutic applications of microglia receptors in Alzheimer's disease.Front Immunol. 2025 Feb 14;16:1508023. doi: 10.3389/fimmu.2025.1508023. eCollection 2025. Front Immunol. 2025. PMID: 40028337 Free PMC article. Review.
-
Oleoylethanolamide facilitates PPARα and TFEB signaling and attenuates Aβ pathology in a mouse model of Alzheimer's disease.Mol Neurodegener. 2023 Aug 15;18(1):56. doi: 10.1186/s13024-023-00648-x. Mol Neurodegener. 2023. PMID: 37580742 Free PMC article.
-
Behaviour Hallmarks in Alzheimer's Disease 5xFAD Mouse Model.Int J Mol Sci. 2024 Jun 20;25(12):6766. doi: 10.3390/ijms25126766. Int J Mol Sci. 2024. PMID: 38928472 Free PMC article. Review.
-
Exploring the molecular mechanisms underlying neuroprotective effect of ellagic acid in okadaic acid-induced Alzheimer's phenotype.Metab Brain Dis. 2024 Oct;39(7):1417-1432. doi: 10.1007/s11011-024-01405-9. Epub 2024 Aug 12. Metab Brain Dis. 2024. PMID: 39133454
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

