Transition Metal-Free Regio- and Stereo-Selective trans Hydroboration of 1,3-Diynes: A Phosphine-Catalyzed Access to (E)-1-Boryl-1,3-Enynes
- PMID: 35917135
- PMCID: PMC9804376
- DOI: 10.1002/chem.202202349
Transition Metal-Free Regio- and Stereo-Selective trans Hydroboration of 1,3-Diynes: A Phosphine-Catalyzed Access to (E)-1-Boryl-1,3-Enynes
Abstract
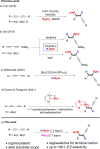
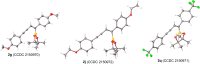
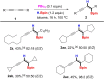
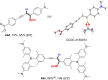
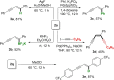
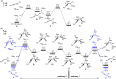
We report a transition metal-free, regio- and stereo-selective, phosphine-catalyzed method for the trans hydroboration of 1,3-diynes with pinacolborane that affords (E)-1-boryl-1,3-enynes. The reaction proceeds with excellent selectivity for boron addition to the external carbon of the 1,3-diyne framework as unambiguously established by NMR and X-ray crystallographic studies. The reaction displays a broad substrate scope including unsymmetrical diynes to generate products in high yield (up to 95 %). Experimental and theoretical studies suggest that phosphine attack on the alkyne is a key process in the catalytic cycle.
Keywords: enediyne; enyne; hydroboration; organocatalytic; stereoselective.
© 2022 The Authors. Chemistry - A European Journal published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Chemo-, Regio-, and Stereoselective cis-Hydroboration of 1,3-Enynes: Copper-Catalyzed Access to (Z,Z)- and (Z,E)-2-Boryl-1,3-dienes.Org Lett. 2024 Jul 26;26(29):6136-6141. doi: 10.1021/acs.orglett.4c01929. Epub 2024 Jul 17. Org Lett. 2024. PMID: 39018130 Free PMC article.
-
Mechanism and Origin of the Stereoselectivity in the Palladium-Catalyzed trans Hydroboration of Internal 1,3-Enynes with an Azaborine-Based Phosphine Ligand.Chemistry. 2018 Jan 2;24(1):178-186. doi: 10.1002/chem.201704035. Epub 2017 Dec 4. Chemistry. 2018. PMID: 29068105
-
Transition Metal-Free Trans Hydroboration of Alkynoic Acid Derivatives: Experimental and Theoretical Studies.J Org Chem. 2018 Sep 7;83(17):10436-10444. doi: 10.1021/acs.joc.8b01493. Epub 2018 Jul 20. J Org Chem. 2018. PMID: 29986587
-
Rhodium-Catalyzed (5 + 2) and (5 + 1) Cycloadditions Using 1,4-Enynes as Five-Carbon Building Blocks.Acc Chem Res. 2020 Jan 21;53(1):231-243. doi: 10.1021/acs.accounts.9b00477. Epub 2019 Dec 10. Acc Chem Res. 2020. PMID: 31820914 Free PMC article. Review.
-
Hydroelementation of diynes.Chem Soc Rev. 2022 Feb 7;51(3):869-994. doi: 10.1039/d1cs00485a. Chem Soc Rev. 2022. PMID: 35005762 Review.
Cited by
-
Ligand-controlled regiodivergent Ni-catalyzed trans-hydroboration/carboboration of internal alkynes with B2pin2.Chem Sci. 2024 Jan 4;15(6):2236-2242. doi: 10.1039/d3sc04184k. eCollection 2024 Feb 7. Chem Sci. 2024. PMID: 38332812 Free PMC article.
-
An easy-to-perform evaluation of steric properties of Lewis acids.Chem Sci. 2023 Feb 6;14(9):2275-2288. doi: 10.1039/d3sc00037k. eCollection 2023 Mar 1. Chem Sci. 2023. PMID: 36873848 Free PMC article.
-
Nickel boryl complexes and nickel-catalyzed alkyne borylation.Chem Sci. 2023 Jan 27;14(8):2215-2228. doi: 10.1039/d2sc04690c. eCollection 2023 Feb 22. Chem Sci. 2023. PMID: 36845942 Free PMC article.
-
Chemo-, Regio-, and Stereoselective cis-Hydroboration of 1,3-Enynes: Copper-Catalyzed Access to (Z,Z)- and (Z,E)-2-Boryl-1,3-dienes.Org Lett. 2024 Jul 26;26(29):6136-6141. doi: 10.1021/acs.orglett.4c01929. Epub 2024 Jul 17. Org Lett. 2024. PMID: 39018130 Free PMC article.
-
Mechanism of Pd/Senphos-Catalyzed trans-Hydroboration of 1,3-Enynes: Experimental and Computational Evidence in Support of the Unusual Outer-Sphere Oxidative Addition Pathway.J Org Chem. 2023 Feb 17;88(4):2415-2424. doi: 10.1021/acs.joc.2c02841. Epub 2023 Feb 8. J Org Chem. 2023. PMID: 36752741 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources

