Facile production of quercetin nanoparticles using 3D printed centrifugal flow reactors
- PMID: 35919149
- PMCID: PMC9295137
- DOI: 10.1039/d2ra02745c
Facile production of quercetin nanoparticles using 3D printed centrifugal flow reactors
Abstract
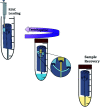
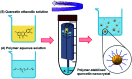
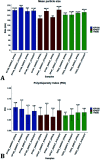
Drug nanocrystals are a delivery system comprised of an active pharmaceutical ingredient, with small amounts of a surface stabilizer. Despite offering simplicity in formulation, their manufacture can be a challenging endeavour; this is especially true when the production is performed using microfluidic devices. Although precipitation within microchannels can lead to issues such as clogging, microfluidics is an appealing manufacturing method as it provides fine control over mixing conditions. This allows production of nanoparticles with a narrower size distribution and greater reproducibility compared to batch methods. To generate microfluidic devices cost effectively, replica moulding techniques are considered the manufacturing standard. Due to its simplicity and relatively low cost, 3D printing has become prevalent at the laboratory scale, especially during iterative development of new devices. A challenge of microfluidic-based methods is that they require specialized equipment and multi-step procedures, making them less accessible to users with no previous experience. In a recent study we developed a 3D printed flow-through reactor, referred to as reactor-in-a-centrifuge (RIAC). It is a simple device designed to fit in a 50 mL tube and actuated using a laboratory centrifuge, which removes the need for specialized instrumentation. The manufacturing capabilities of the RIAC have been already proven, by reproducible production of liposomes and silver nanoparticles. The present work demonstrates the use of RIACs with a straight- and spiral-shaped channel architecture to produce quercetin nanocrystals, with therapeutically relevant size (190-302 nm) and very low size dispersity (polydispersity index, PDI < 0.1). The work focused on evaluating how changes in operational parameters (actuation speed) and formulation components (medium viscosity and stabilizer type), impacted on nanocrystal size and PDI. Under all tested conditions the obtained nanocrystals had a smaller size and narrower size distribution, when compared to those produced with alternative methods. The obtained quercetin nanosuspensions however showed limited stability, which should be addressed in future investigations. The simplicity of the RIAC makes it an appealing technology to research groups, especially in low-resource settings and without prior expertise in microfluidics.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures








Similar articles
-
Rapid Production of Nanoscale Liposomes Using a 3D-Printed Reactor-In-A-Centrifuge: Formulation, Characterisation, and Super-Resolution Imaging.Micromachines (Basel). 2023 Sep 12;14(9):1763. doi: 10.3390/mi14091763. Micromachines (Basel). 2023. PMID: 37763926 Free PMC article.
-
Manufacturing of 3D-Printed Microfluidic Devices for the Synthesis of Drug-Loaded Liposomal Formulations.Int J Mol Sci. 2021 Jul 28;22(15):8064. doi: 10.3390/ijms22158064. Int J Mol Sci. 2021. PMID: 34360832 Free PMC article.
-
Three-Dimensional-Printed Vortex Tube Reactor for Continuous Flow Synthesis of Polyglycolic Acid Nanoparticles with High Productivity.Nanomaterials (Basel). 2023 Sep 29;13(19):2679. doi: 10.3390/nano13192679. Nanomaterials (Basel). 2023. PMID: 37836320 Free PMC article.
-
Merits and advances of microfluidics in the pharmaceutical field: design technologies and future prospects.Drug Deliv. 2022 Dec;29(1):1549-1570. doi: 10.1080/10717544.2022.2069878. Drug Deliv. 2022. PMID: 35612293 Free PMC article. Review.
-
Microfluidics: a transformational tool for nanomedicine development and production.J Drug Target. 2016 Nov;24(9):821-835. doi: 10.1080/1061186X.2016.1198354. Epub 2016 Aug 5. J Drug Target. 2016. PMID: 27492254 Review.
Cited by
-
Polymeric Systems for the Controlled Release of Flavonoids.Pharmaceutics. 2023 Feb 13;15(2):628. doi: 10.3390/pharmaceutics15020628. Pharmaceutics. 2023. PMID: 36839955 Free PMC article. Review.
-
Rapid Production of Nanoscale Liposomes Using a 3D-Printed Reactor-In-A-Centrifuge: Formulation, Characterisation, and Super-Resolution Imaging.Micromachines (Basel). 2023 Sep 12;14(9):1763. doi: 10.3390/mi14091763. Micromachines (Basel). 2023. PMID: 37763926 Free PMC article.
References
LinkOut - more resources
Full Text Sources

