The protective effect of DNA aptamer on osteonecrosis of the femoral head by alleviating TNF-α-mediated necroptosis via RIP1/RIP3/MLKL pathway
- PMID: 35919280
- PMCID: PMC9307900
- DOI: 10.1016/j.jot.2022.07.001
The protective effect of DNA aptamer on osteonecrosis of the femoral head by alleviating TNF-α-mediated necroptosis via RIP1/RIP3/MLKL pathway
Abstract
Background: The process of necroptosis mediated by tumor necrosis factor alpha (TNF-α) might play an important role in the onset and development of the osteonecrosis of the femoral head (ONFH). The dysfunctions of bone microvascular endothelial cells (BMECs) have been identified as an important part of pathological processes in the steroid-induced ONFH. An aptamer is a single-stranded DNA or RNA oligonucleotide sequence. Previous studies have designed or screened various aptamers that could bind to specific targets or receptors in order to block their effects.
Objective: There are two main objectives in this study: 1) to establish a TNF-α -induced ONFH model in human BMECs in vitro, 2) to verify the effects of the TNF-α aptamer (AptTNF-α) on blocking TNF-α activity in the ONFH model.
Methods: Clinical samples were collected for Hematoxylin and Eosin (HE) staining, immunohistochemistry and further BMEC isolation. After cell culture and identification, the cell viability of BMECs after incubation with TNF-α was assessed by Cell Counting Kit-8 (CCK8). The necroptosis of BMECs was detected by the TUNEL and Annexin V-FITC/PI staining. The attenuation of TNF-α cytotoxicity by AptTNF-α was evaluated by CCK8 at first. Then, the molecular mechanism was explored by the quantitative real-time polymerase chain reaction and western blotting.
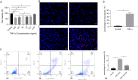
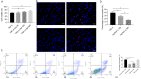
Results: The expression level of TNF-α was significantly up-regulated in bone tissues of ONFH patients. The identification of BMECs was verified by the high expressions of CD31 and vWF. Results from CCK8, TUNEL staining and Annexin V-FITC/PI assay demonstrated reduced cell viability and increased necroptosis of BMECs after TNF-α stimulation. Further investigations showed that TNF-α cytotoxicity could be attenuated by the AptTNF-α in a dose-dependent manner. Necroptosis mediated by TNF-α in the ONFH model was regulated by the receptor-interacting protein kinase 1 (RIPK1)/receptor-interacting protein kinase 3 (RIPK3)/mixed lineage kinase domain-like protein (MLKL) signalling pathway.
Conclusion: We established a TNF-α-induced ONFH model in human BMECs in vitro. Our study also demonstrated that the AptTNF-α could protect BMECs from necroptosis by inhibiting the RIP1/RIP3/MLKL signalling pathway.The Translational Potential of this Article: The effective protection from cell necroptosis provided by the DNA aptamer demonstrated its translational potential as a new type of TNF-α inhibitor in clinical treatments for patients with ONFH.
Keywords: Aptamer; Bone microvascular endothelial cell; Necroptosis; Osteonecrosis of the femoral head; TNF-α.
© 2022 The Authors.
Conflict of interest statement
The authors have no potential financial and non-financial conflicts of interest relevant to this article.
Figures



Similar articles
-
Luteolin ameliorates necroptosis in Glucocorticoid-induced osteonecrosis of the femoral head via RIPK1/RIPK3/MLKL pathway based on network pharmacology analysis.Biochem Biophys Res Commun. 2023 Jun 18;661:108-118. doi: 10.1016/j.bbrc.2023.04.023. Epub 2023 Apr 11. Biochem Biophys Res Commun. 2023. PMID: 37099894
-
Establishment of the multi-component bone-on-a-chip: to explore therapeutic potential of DNA aptamers on endothelial cells.Front Cell Dev Biol. 2023 Jun 12;11:1183163. doi: 10.3389/fcell.2023.1183163. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37377731 Free PMC article.
-
Panax notoginseng Saponins Protect Brain Microvascular Endothelial Cells against Oxygen-Glucose Deprivation/Resupply-Induced Necroptosis via Suppression of RIP1-RIP3-MLKL Signaling Pathway.Neurochem Res. 2022 Nov;47(11):3261-3271. doi: 10.1007/s11064-022-03675-0. Epub 2022 Jul 29. Neurochem Res. 2022. PMID: 35904697
-
Necroptosis in health and diseases.Semin Cell Dev Biol. 2014 Nov;35:14-23. doi: 10.1016/j.semcdb.2014.07.013. Epub 2014 Aug 1. Semin Cell Dev Biol. 2014. PMID: 25087983 Review.
-
Molecular mechanisms of necroptosis and relevance for neurodegenerative diseases.Int Rev Cell Mol Biol. 2020;353:31-82. doi: 10.1016/bs.ircmb.2019.12.006. Epub 2020 Feb 13. Int Rev Cell Mol Biol. 2020. PMID: 32381178 Review.
Cited by
-
Unraveling the Role of Endothelial Dysfunction in Osteonecrosis of the Femoral Head: A Pathway to New Therapies.Biomedicines. 2024 Mar 15;12(3):664. doi: 10.3390/biomedicines12030664. Biomedicines. 2024. PMID: 38540277 Free PMC article. Review.
-
JianPiYiShen formula prevents cisplatin-induced acute kidney injury in mice by improving necroptosis through MAPK pathway.BMC Complement Med Ther. 2024 Feb 24;24(1):101. doi: 10.1186/s12906-024-04366-9. BMC Complement Med Ther. 2024. PMID: 38402163 Free PMC article.
-
Bone and the Unfolded Protein Response: In Sickness and in Health.Calcif Tissue Int. 2023 Jul;113(1):96-109. doi: 10.1007/s00223-023-01096-x. Epub 2023 May 27. Calcif Tissue Int. 2023. PMID: 37243756 Free PMC article. Review.
-
Aucubin suppresses TLR4/NF-κB signalling to shift macrophages toward M2 phenotype in glucocorticoid-associated osteonecrosis of the femoral head.J Cell Mol Med. 2024 Aug;28(15):e18583. doi: 10.1111/jcmm.18583. J Cell Mol Med. 2024. PMID: 39123292 Free PMC article.
-
Bioinformatics analysis of PANoptosis regulators in the diagnosis and subtyping of steroid-induced osteonecrosis of the femoral head.Medicine (Baltimore). 2024 May 3;103(18):e37837. doi: 10.1097/MD.0000000000037837. Medicine (Baltimore). 2024. PMID: 38701259 Free PMC article.
References
-
- Barquet A., Mayora G., Guimaraes J.M., Suárez R., Giannoudis P.V. Avascular necrosis of the femoral head following trochanteric fractures in adults: a systematic review. Injury. 2014;45:1848–1858. - PubMed
-
- Guerado E., Caso E. The physiopathology of avascular necrosis of the femoral head: an update. Injury. 2016;47:S16–S26. - PubMed
-
- Zheng L.W., Wang W.C., Mao X.Z., Luo Y.H., Tong Z.Y., Li D. TNF-α regulates the early development of avascular necrosis of the femoral head by mediating osteoblast autophagy and apoptosis via the p38 MAPK/NF-κB signalling pathway. Cell Biol Int. 2020;44:1881–1889. - PubMed
-
- Degterev A., Huang Z., Boyce M., Li Y., Jagtap P., Mizushima N., et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat Chem Biol. 2005;1:112–119. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

