Early recognition and treatment of pre-VITT syndrome after adenoviral vector-based SARS-CoV-2 vaccination may prevent from thrombotic complications: review of published cases and clinical pathway
- PMID: 35919343
- PMCID: PMC9242075
- DOI: 10.1093/ehjopen/oeac036
Early recognition and treatment of pre-VITT syndrome after adenoviral vector-based SARS-CoV-2 vaccination may prevent from thrombotic complications: review of published cases and clinical pathway
Abstract
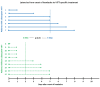
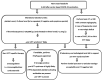
Vaccine-induced immune thrombotic thrombocytopenia (VITT) is a rare but highly morbid complication after adenoviral vector-based SARS-CoV-2 vaccination. The pre-VITT syndrome is defined as vaccine-induced immune thrombocytopenia without thrombosis typically presenting with new-onset headache. This review aims to identify at-risk patients before complications such as cerebral venous sinus thrombosis occur. We review previously published reports of 19 patients (median age 35 years, range 23-74; 16 females) who met the diagnostic criteria for a pre-VITT syndrome. Seven patients progressed to VITT, 12 patients did not. Patients who experienced VITT received delayed treatment. The median interval between the onset of headache and VITT-treatment (i.e. anticoagulation, immune globulins, or corticosteroids) was 5 days (range 1-8 days) compared with 2 days (0-5 days) in those without subsequent VITT (P = 0.033). The interval from onset of headache to anticoagulation was longer in patients with VITT (median 7 vs. 2 days; range 3-9 vs. 0-7 days; P = 0.01). Anticoagulation was safe in all patients with a pre-VITT syndrome as no haemorrhagic complications occurred after anticoagulation was started despite low platelets. The transient decline of platelet count after admission was significantly more pronounced in patients who progressed to VITT (median 67 vs. 0 × 103/µL; range 0-77 × 103/µL vs. 0-10 × 103/µL; P = 0.005). d-dimers did not differ between groups. Pre-VITT syndrome is a 'red flag' and allows to identify and preemptively treat patients at-risk of further progression to VITT. However, it must be distinguished from post-vaccination immune thrombocytopenia.
Keywords: Anticoagulation; Headache; Pre-VITT syndrome; TTS; Thrombocytopenia; VITT.
© The Author(s) 2022. Published by Oxford University Press on behalf of the European Society of Cardiology.
Figures


Similar articles
-
Retrospective review COVID-19 vaccine induced thrombotic thrombocytopenia and cerebral venous thrombosis-what can we learn from the immune response.Clin Imaging. 2022 Oct;90:63-70. doi: 10.1016/j.clinimag.2022.06.020. Epub 2022 Jul 15. Clin Imaging. 2022. PMID: 35926315 Free PMC article.
-
Vaccine induced thrombotic thrombocytopenia: The shady chapter of a success story.Metabol Open. 2021 Sep;11:100101. doi: 10.1016/j.metop.2021.100101. Epub 2021 Jun 18. Metabol Open. 2021. PMID: 34179744 Free PMC article.
-
Late-Onset Vaccine-Induced Immune Thombotic Thrombocytopenia (VITT) with Cerebral Venous Sinus Thrombosis.J Stroke Cerebrovasc Dis. 2022 Apr;31(4):106311. doi: 10.1016/j.jstrokecerebrovasdis.2022.106311. Epub 2022 Jan 29. J Stroke Cerebrovasc Dis. 2022. PMID: 35093626 Free PMC article.
-
Vaccine-induced immune thrombotic thrombocytopenia.Best Pract Res Clin Haematol. 2022 Sep;35(3):101381. doi: 10.1016/j.beha.2022.101381. Epub 2022 Sep 13. Best Pract Res Clin Haematol. 2022. PMID: 36494147 Free PMC article. Review.
-
Ischemic Stroke and Vaccine-Induced Immune Thrombotic Thrombocytopenia following COVID-19 Vaccine: A Case Report with Systematic Review of the Literature.Cerebrovasc Dis. 2022;51(6):722-734. doi: 10.1159/000524290. Epub 2022 May 5. Cerebrovasc Dis. 2022. PMID: 35512656 Free PMC article.
Cited by
-
Incidence and management of the main serious adverse events reported after COVID-19 vaccination.Pharmacol Res Perspect. 2024 Jun;12(3):e1224. doi: 10.1002/prp2.1224. Pharmacol Res Perspect. 2024. PMID: 38864106 Free PMC article. Review.
-
Vaccine-induced immune thrombotic thrombocytopenia: what do we know hitherto?Front Med (Lausanne). 2023 May 16;10:1155727. doi: 10.3389/fmed.2023.1155727. eCollection 2023. Front Med (Lausanne). 2023. PMID: 37261122 Free PMC article. Review.
-
Differences and similarities between COVID-19 related-headache and COVID-19 vaccine related-headache. A case-control study.Rev Neurol. 2023 Nov 16;77(10):229-239. doi: 10.33588/rn.7710.2023063. Rev Neurol. 2023. PMID: 37962534 Free PMC article. English, Spanish.
-
Highlights from 2022 in EHJ Open.Eur Heart J Open. 2022 Dec 30;2(6):oeac084. doi: 10.1093/ehjopen/oeac084. eCollection 2022 Nov. Eur Heart J Open. 2022. PMID: 36600883 Free PMC article. No abstract available.
-
Vaccination-induced thrombocytopenia and thrombosis (VITT) and pre-VITT: Do not miss (or misdiagnose) the new member of the thrombotic thrombocytopenias family.Eur Heart J Open. 2022 Sep 7;2(5):oeac056. doi: 10.1093/ehjopen/oeac056. eCollection 2022 Sep. Eur Heart J Open. 2022. PMID: 36156870 Free PMC article. No abstract available.
References
-
- World Health Organization . Guidance for clinical case management of thrombosis with thrombocytopenia syndrome (TTS) following vaccination to prevent coronavirus disease (COVID-19). https://www.who.int/publications/i/item/WHO-2019-nCoV-TTS-2021.1 (20 February 2022).
-
- van de Munckhof A, Krzywicka K, de Sousa DA, van Kammen MS, Heldner MR, Jood K, Lindgren E, Tatlisumak T, Putaala J, Kremer Hovinga JA, Middeldorp S, Levi M, Arnold M, Ferro JM, Coutinho JM. Declining mortality of cerebral venous sinus thrombosis with thrombocytopenia after SARS-CoV-2 vaccination. Eur J Neurol 2022;29:339–344. - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

