Modeling propofol-induced cardiotoxicity in the isolated-perfused newborn mouse heart
- PMID: 35923108
- PMCID: PMC9350423
- DOI: 10.14814/phy2.15402
Modeling propofol-induced cardiotoxicity in the isolated-perfused newborn mouse heart
Abstract
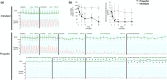
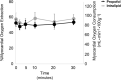
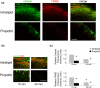
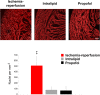
Infants and children are vulnerable to developing propofol infusion syndrome (PRIS) and young age is a risk factor. Cardiac involvement is often prominent and associated with death. However, the mechanisms of pediatric PRIS are poorly understood because of the paucity of investigation and lack of a gold standard animal model. Unfortunately, in vivo modeling of PRIS in a newborn mouse is not feasible and would be complicated by confounders. Thus, we focused on propofol-induced cardiotoxicity and aimed to develop an ex-vivo model in the isolated-perfused newborn mouse heart. We hypothesized that the model would recapitulate the key cardiac features of PRIS seen in infants and children and would corroborate prior in vitro observations. Isolated perfused newborn mouse hearts were exposed to a toxic dose of propofol or intralipid for 30-min. Surface electrocardiogram, ventricular contractile force, and oxygen extraction were measured over time. Real-time multiphoton laser imaging was utilized to quantify calcein and tetramethylrhodamine ethyl ester fluorescence. Propidium iodide uptake was assessed following drug exposure. A toxic dose of propofol rapidly induced dysrhythmias, depressed ventricular contractile function, impaired the mitochondrial membrane potential, and increased open probability of the permeability transition pore in propofol-exposed hearts without causing cell death. These features mimicked the hallmarks of pediatric PRIS and corroborated prior observations made in isolated newborn cardiomyocyte mitochondria. Thus, acute propofol-induced cardiotoxicity in the isolated-perfused developing mouse heart may serve as a relevant ex-vivo model for pediatric PRIS.
Keywords: Langendorff preparation; cardiotoxicity; heart; isolated-perfused heart; model; newborn; propofol; propofol infusion syndrome.
© 2022 The Authors. Physiological Reports published by Wiley Periodicals LLC on behalf of The Physiological Society and the American Physiological Society.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Propofol toxicity in the developing mouse heart mitochondria.Pediatr Res. 2022 Nov;92(5):1341-1349. doi: 10.1038/s41390-022-01985-1. Epub 2022 Feb 16. Pediatr Res. 2022. PMID: 35173299 Free PMC article.
-
Propofol infusion syndrome: a structured review of experimental studies and 153 published case reports.Crit Care. 2015 Nov 12;19:398. doi: 10.1186/s13054-015-1112-5. Crit Care. 2015. PMID: 26558513 Free PMC article. Review.
-
Incidence of propofol-related infusion syndrome in critically ill adults: a prospective, multicenter study.Crit Care. 2009;13(5):R169. doi: 10.1186/cc8145. Epub 2009 Oct 29. Crit Care. 2009. PMID: 19874582 Free PMC article.
-
Propofol infusion syndrome: an overview of a perplexing disease.Drug Saf. 2008;31(4):293-303. doi: 10.2165/00002018-200831040-00003. Drug Saf. 2008. PMID: 18366240 Review.
-
The incidence of propofol infusion syndrome in critically-ill patients.J Crit Care. 2022 Oct;71:154098. doi: 10.1016/j.jcrc.2022.154098. Epub 2022 Jun 17. J Crit Care. 2022. PMID: 35724444
Cited by
-
Toxic effects of prolonged propofol exposure on cardiac development in zebrafish larvae.BMC Anesthesiol. 2025 Feb 18;25(1):81. doi: 10.1186/s12871-025-02942-1. BMC Anesthesiol. 2025. PMID: 39966746 Free PMC article.
-
Effects of the Quinone Analog Ubiquinone-5 on Murine Mitochondria and Hypnosis.Anesthesiology. 2025 Sep 1;143(3):641-660. doi: 10.1097/ALN.0000000000005549. Epub 2025 May 2. Anesthesiology. 2025. PMID: 40315373
-
A primordial target: Mitochondria mediate both primary and collateral anesthetic effects of volatile anesthetics.Exp Biol Med (Maywood). 2023 Apr;248(7):545-552. doi: 10.1177/15353702231165025. Epub 2023 May 19. Exp Biol Med (Maywood). 2023. PMID: 37208922 Free PMC article. Review.
-
Sulfide quinone oxidoreductase contributes to voltage sensing of the mitochondrial permeability transition pore.FASEB J. 2024 Feb 29;38(4):e23494. doi: 10.1096/fj.202301280R. FASEB J. 2024. PMID: 38376922 Free PMC article.
-
Ferroptosis-induced Cardiotoxicity and Antitumor Drugs.Curr Med Chem. 2024;31(31):4935-4957. doi: 10.2174/0929867331666230719124453. Curr Med Chem. 2024. PMID: 37469161 Review.
References
-
- Bray, R. J. (1998). Propofol infusion syndrome in children. Paediatric Anaesthesia, 8, 491–499. - PubMed
-
- Bray, R. J. (2002). The propofol infusion syndrome in infants and children: Can we predict the risk? Current Opinion in Anaesthesiology, 15, 339–342. - PubMed
-
- Campos, S. , Monteiro, J. , Valenzuela, B. , Gonçalinho, H. , de Pinho, P. G. , Fresco, P. , Félix, L. , & Antunes, L. (2016). Evidence of different propofol pharmacokinetics under short and prolonged infusion times in rabbits. Basic & Clinical Pharmacology & Toxicology, 118, 421–431. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

