A Novel Delivery System of RGD-HSA Loaded GEM/CUR Nanoparticles for the Treatment of Pancreatic Cancer Therapy
- PMID: 35923931
- PMCID: PMC9339944
- DOI: 10.2147/DDDT.S366558
A Novel Delivery System of RGD-HSA Loaded GEM/CUR Nanoparticles for the Treatment of Pancreatic Cancer Therapy
Abstract
Introduction: Pancreatic cancer is one of the most common malignant tumors and is characterized by high malignancy, occult incidence and poor prognosis. Traditional chemotherapy drugs have limited efficacy and strong side effects. Therefore, there is an urgent need for a better treatment of the malignancy.
Methods: The prepared arginine glycine peptide (RGD)-human serum albumin (HSA)-Gemcitabine (GEM)/Curcumin (CUR) nanoparticles (NPs) were characterized for physicochemical properties, stability and in vitro release. Comparisons of HSA-GEM/CUR NPs and RGD-HSA-GEM/CUR NPs regarding tissue distributions and pharmacodynamics were also carried out using mice as the animal models.
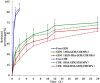
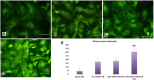
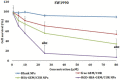
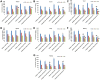
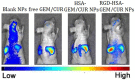
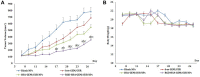
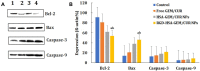
Results: Transmission electron micrographs showed that RGD peptide-conjugated HSA-NPs had an irregular surface, good dispersion (PDI=0.139±0.03) and a uniform size distribution (Mean PS=115.6±5.7 nm). The ζ-potential was -17.3 mV. As regards in vitro release, non RGD modified NPs showed a faster release rate in 24 hours, yielding a release amount of 75% for GEM and 72% for CUR. RGD-HSA-GEM/CUR NPs exhibited 67% of accumulated release of GEM (63% for CUR) in 24 hours. This may be due to the HSA chain covering the surface of NPs, which hindered the drug release. The cytotoxicity of GEM/CUR co-loaded NPs was significantly higher than that of single-drug NPs (P < 0.05). In vivo study results indicated that RGD-HSA-GEM/CUR NPs had notable targeting effect on subcutaneous tumors, with a potential to actively deliver drugs to tumor tissues.
Conclusion: In this study, we prepared RGD-HSA-GEM/CUR NPs that had both good water solubility and tumor-targeting property. The results also showed that the RGD modified NPs had advantages in increasing GEM/CUR concentration at tumor sites and reducing its distribution in peripheral organs.
Keywords: Gemcitabine/Curcumin; arginine glycine peptide; human serum albumin; nanoparticles; pancreatic cancer.
© 2022 Ma et al.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

