A Tissue-Tended Mycophenolate-Modified Nanoparticle Alleviates Systemic Lupus Erythematosus in MRL/ Lpr Mouse Model Mainly by Promoting Local M2-Like Macrophagocytes Polarization
- PMID: 35924257
- PMCID: PMC9342721
- DOI: 10.2147/IJN.S361400
A Tissue-Tended Mycophenolate-Modified Nanoparticle Alleviates Systemic Lupus Erythematosus in MRL/ Lpr Mouse Model Mainly by Promoting Local M2-Like Macrophagocytes Polarization
Abstract
Background: Mycophenolate mofetil (MMF), for which the bioactive metabolite is mycophenolic acid (MPA), is a frequently used immunosuppressant for systemic lupus erythematosus (SLE). However, its short half-life and poor biodistribution into cells and tissues hinder its clinical efficacy. Our dextran mycophenolate-based nanoparticles (MPA@Dex-MPA NPs) have greatly improved the pharmacokinetics of MMF/MPA. We here tested the therapeutic efficacy of MPA@Dex-MPA NPs against SLE and investigated the underlying mechanism.
Methods: The tissue and immune cell biodistributions of MPA@Dex-MPA NPs were traced using live fluorescence imaging system and flow cytometry, respectively. Serological proinflammatory mediators and kidney damage were detected to assess the efficacy of MPA@Dex-MPA NPs treatments of MRL/lpr lupus-prone mice. Immune cell changes in the kidney and spleen were further analyzed post-treatment via flow cytometry. Bone marrow-derived macrophages were used to investigate the potential mechanism.
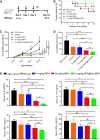
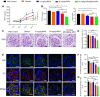
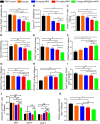
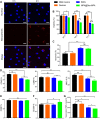
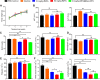
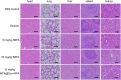
Results: MPA@Dex-MPA NPs exhibited superior therapeutic efficacy and safety in the MRL/lpr mice using significantly lower administration dosage (one-fifth) and frequency (once/3 days) compared to MMF/MPA used in ordinary practice. The overall prognosis of the mice was improved as they showed lower levels of serological proinflammatory mediators. Moreover, kidney injury was alleviated with reduced pathological signs and decreased urine protein-creatinine ratio. Further investigations of the underlying mechanism revealed a preferential penetration and persistent retention of MPA@Dex-MPA NPs in the spleen and kidney, where they were mostly phagocytosed by macrophages. The macrophages were found to be polarized towards a CD206+ M2-like phenotype, with a downregulation of surface CD80 and CD40, and reduced TNF-α production in the spleen and kidney and in vitro. The expansion of T cells was also significantly inhibited in these two organs.
Conclusion: Our research improved the efficacy of MPA for MRL/lpr mice through synthesizing MPA@Dex-MPA NPs to enhance its tissue biodistribution and explored the possible mechanism, providing a promising strategy for SLE therapy.
Keywords: macrophage polarization; mycophenolate; nanoparticles; systemic lupus erythematosus; tissue damage.
© 2022 Jiang et al.
Conflict of interest statement
The authors report no conflicts of interest in relation to this study.
Figures


Similar articles
-
Mesenchymal Stem Cells-derived Exosomes Ameliorate Lupus by Inducing M2 Macrophage Polarization and Regulatory T Cell Expansion in MRL/lpr Mice.Immunol Invest. 2022 Aug;51(6):1785-1803. doi: 10.1080/08820139.2022.2055478. Epub 2022 Mar 25. Immunol Invest. 2022. PMID: 35332841
-
Modulation of intra-pulmonary TGF-beta expression by mycophenolate mofetil in lupus prone MRL/lpr mice.Lupus. 2005;14(8):583-92. doi: 10.1191/0961203305lu2170oa. Lupus. 2005. PMID: 16175929
-
Effect of mycophenolate mofetil on severity of nephritis and nitric oxide production in lupus-prone MRL/lpr mice.Lupus. 2002;11(7):411-8. doi: 10.1191/0961203302lu214oa. Lupus. 2002. PMID: 12195781
-
METTL3 facilitates kidney injury through promoting IRF4-mediated plasma cell infiltration via an m6A-dependent manner in systemic lupus erythematosus.BMC Med. 2024 Nov 5;22(1):511. doi: 10.1186/s12916-024-03735-y. BMC Med. 2024. PMID: 39501302 Free PMC article.
-
Mycophenolate mofetil and systemic lupus erythematosus: an overview.Lupus. 2005;14 Suppl 1:s9-11. doi: 10.1191/0961203305lu2111oa. Lupus. 2005. PMID: 15803925 Review.
Cited by
-
Multiple Shades of Gray-Macrophages in Acute Allograft Rejection.Int J Mol Sci. 2023 May 4;24(9):8257. doi: 10.3390/ijms24098257. Int J Mol Sci. 2023. PMID: 37175964 Free PMC article. Review.
-
The applications of functional materials-based nano-formulations in the prevention, diagnosis and treatment of chronic inflammation-related diseases.Front Pharmacol. 2023 Aug 1;14:1222642. doi: 10.3389/fphar.2023.1222642. eCollection 2023. Front Pharmacol. 2023. PMID: 37593176 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

