Streptococcus pneumoniae Rapidly Translocate from the Nasopharynx through the Cribriform Plate to Invade the Outer Meninges
- PMID: 35924840
- PMCID: PMC9426477
- DOI: 10.1128/mbio.01024-22
Streptococcus pneumoniae Rapidly Translocate from the Nasopharynx through the Cribriform Plate to Invade the Outer Meninges
Abstract
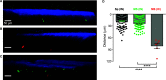
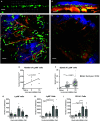
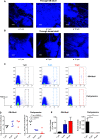
The entry routes and translocation mechanisms of microorganisms or particulate materials into the central nervous system remain obscure We report here that Streptococcus pneumoniae (pneumococcus), or polystyrene microspheres of similar size, appear in the meninges of the dorsal cortex of mice within minutes of inhaled delivery. Recovery of viable bacteria from dissected tissue and fluorescence microscopy show that up to at least 72 h, pneumococci and microspheres were predominantly found in the outer of the two meninges: the pachymeninx. No pneumococci were found in blood or cerebrospinal fluid. Intravital imaging through the skull, aligned with flow cytometry showed recruitment and activation of LysM+ cells in the dorsal pachymeninx at 5 and 10 hours following intranasal infection. Imaging of the cribriform plate suggested that both pneumococci and microspheres entered through the foramina via an inward flow of fluid connecting the nose to the pachymeninx. Our findings bring new insight into the varied mechanisms of pneumococcal invasion of the central nervous system, but they are also pertinent to the delivery of drugs to the brain and the entry of airborne particulate matter into the cranium. IMPORTANCE Using two-photon imaging, we show that pneumococci translocate from the nasopharynx to the dorsal meninges of a mouse in the absence of any bacteria found in blood or cerebrospinal fluid. Strikingly, this takes place within minutes of inhaled delivery of pneumococci, suggesting the existence of an inward flow of fluid connecting the nasopharynx to the meninges, rather than a receptor-mediated mechanism. We also show that this process is size dependent, as microspheres of the same size as pneumococci can translocate along the same pathway, while larger size microspheres cannot. Furthermore, we describe the host response to invasion of the outer meninges. Our study provides a completely new insight into the key initial events that occur during the translocation of pneumococci directly from the nasal cavity to the meninges, with relevance to the development of intranasal drug delivery systems and the investigations of brain damage caused by inhaled air pollutants.
Keywords: Streptococcus pneumoniae; central nervous system; cribriform plate; infectious disease; inflammation; nose-to-meninges translocation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Jusot J-F, Neill DR, Waters EM, Bangert M, Collins M, Bricio Moreno L, Lawan KG, Moussa MM, Dearing E, Everett DB, Collard J-M, Kadioglu A. 2017. Airborne dust and high temperatures are risk factors for invasive bacterial disease. J Allergy Clin Immunol 139:977–986.e972. doi: 10.1016/j.jaci.2016.04.062. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

