Disentangling personalized treatment effects from "time-of-the-day" confounding in mobile health studies
- PMID: 35925980
- PMCID: PMC9352058
- DOI: 10.1371/journal.pone.0271766
Disentangling personalized treatment effects from "time-of-the-day" confounding in mobile health studies
Abstract
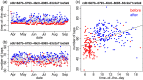
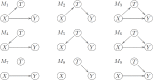
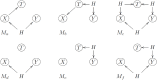
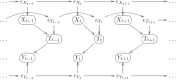
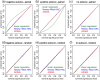
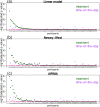
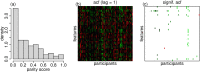
Ideally, a patient's response to medication can be monitored by measuring changes in performance of some activity. In observational studies, however, any detected association between treatment ("on-medication" vs "off-medication") and the outcome (performance in the activity) might be due to confounders. In particular, causal inferences at the personalized level are especially vulnerable to confounding effects that arise in a cyclic fashion. For quick acting medications, effects can be confounded by circadian rhythms and daily routines. Using the time-of-the-day as a surrogate for these confounders and the performance measurements as captured on a smartphone, we propose a personalized statistical approach to disentangle putative treatment and "time-of-the-day" effects, that leverages conditional independence relations spanned by causal graphical models involving the treatment, time-of-the-day, and outcome variables. Our approach is based on conditional independence tests implemented via standard and temporal linear regression models. Using synthetic data, we investigate when and how residual autocorrelation can affect the standard tests, and how time series modeling (namely, ARIMA and robust regression via HAC covariance matrix estimators) can remedy these issues. In particular, our simulations illustrate that when patients perform their activities in a paired fashion, positive autocorrelation can lead to conservative results for the standard regression approach (i.e., lead to deflated true positive detection), whereas negative autocorrelation can lead to anticonservative behavior (i.e., lead to inflated false positive detection). The adoption of time series methods, on the other hand, leads to well controlled type I error rates. We illustrate the application of our methodology with data from a Parkinson's disease mobile health study.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Virtualized clinical studies to assess the natural history and impact of gut microbiome modulation in non-hospitalized patients with mild to moderate COVID-19 a randomized, open-label, prospective study with a parallel group study evaluating the physiologic effects of KB109 on gut microbiota structure and function: a structured summary of a study protocol for a randomized controlled study.Trials. 2021 Apr 2;22(1):245. doi: 10.1186/s13063-021-05157-0. Trials. 2021. PMID: 33810796 Free PMC article.
-
A New Method for Partial Correction of Residual Confounding in Time-Series and Other Observational Studies.Am J Epidemiol. 2017 May 15;185(10):941-949. doi: 10.1093/aje/kwx013. Am J Epidemiol. 2017. PMID: 28430842
-
Instrumental variables and inverse probability weighting for causal inference from longitudinal observational studies.Stat Methods Med Res. 2004 Feb;13(1):17-48. doi: 10.1191/0962280204sm351ra. Stat Methods Med Res. 2004. PMID: 14746439
-
The Evolution of Personalized Behavioral Intervention Technology: Will It Change How We Measure or Deliver Rehabilitation?Stroke. 2017 Aug;48(8):2329-2334. doi: 10.1161/STROKEAHA.117.016620. Epub 2017 Jul 5. Stroke. 2017. PMID: 28679855 Free PMC article. Review. No abstract available.
-
Personalized Technologies in Chronic Gastrointestinal Disorders: Self-monitoring and Remote Sensor Technologies.Clin Gastroenterol Hepatol. 2016 Dec;14(12):1697-1705. doi: 10.1016/j.cgh.2016.05.009. Epub 2016 May 14. Clin Gastroenterol Hepatol. 2016. PMID: 27189911 Free PMC article. Review.
Cited by
-
Impacts on study design when implementing digital measures in Parkinson's disease-modifying therapy trials.Front Digit Health. 2024 Oct 9;6:1430994. doi: 10.3389/fdgth.2024.1430994. eCollection 2024. Front Digit Health. 2024. PMID: 39445101 Free PMC article.
References
-
- Arora S, Venkataraman V, Donohue S, Biglan KM, Dorsey ER, Little MA. High accuracy discrimination of Parkinson’s disease participants from healthy controls using smartphones. IEEE International Conference on Acoustics, Speech and Signal Processing. 2014.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

