Venetoclax ex vivo functional profiling predicts improved progression-free survival
- PMID: 35927247
- PMCID: PMC9352682
- DOI: 10.1038/s41408-022-00710-9
Venetoclax ex vivo functional profiling predicts improved progression-free survival
Conflict of interest statement
PN serves on advisory boards for BMS, Janssen, and Sanofi. NJB receives honoraria from and serves in a consulting or advisory role for Celgene, Janssen, AbbVie, Amgen, Sanofi, and Takeda. MDV serves on advisory boards for Amgen, Janssen, Roche, and Genentech. CCH has received research grants from Oncolytics Biotech, research and personal grants from Janssen, BMS, Sanofi, Nektar, and Karyopharm, and personal grants from Imbrium and Oncopeptides. LTH receives an honorarium from Kite Pharmaceuticals. SL has consulting or advisory roles with Celgene, Bristol-Myers Squibb, Janssen Oncology, Novartis, GlaxoSmithKline, Amgen, AbbVie, Takeda, Merck, Juno Therapeutics, and receives research funding from Celgene, Bristol-Myers Squibb, and Takeda. AKN has a significant financial interest in Janssen pharmaceuticals and has participated on advisory boards and received honoraria from Janssen, Takeda, Amgen, BMS/Celgene, GSK, Oncopeptides, Karyopharm pharmaceuticals, BeyondSprings, Secura Bio, Sanofi, and Adaptive technologies. JLK receives honoraria from Tecnofarma; research support from BMS, Janssen, Bluebird, Sutro Biopharma, Amgen, Abbvie, Fortis Therapeutics, GSK; consultancy for BMS, Janssen; and data safety monitoring board for TG therapeutics. DLJ serves on the advisory board for Stemline Therapeutics. LHB receives research funding, consultancy, and honoraria from AstraZeneca and performs consultancy for Genentech and Abbvie. The remaining authors declare no competing financial interests.
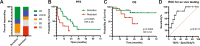
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

