A Linarin Derivative Protects against Ischemia-Induced Neuronal Injury in Mice by Promoting Cerebral Blood Flow Recovery via KDELR-Dependent CSPG4 Activation
- PMID: 35927993
- PMCID: PMC9345725
- DOI: 10.1155/2022/6434086
A Linarin Derivative Protects against Ischemia-Induced Neuronal Injury in Mice by Promoting Cerebral Blood Flow Recovery via KDELR-Dependent CSPG4 Activation
Abstract
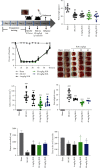
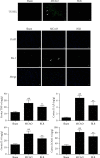
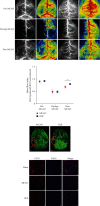
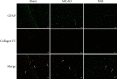
The cerebral ischemic microvascular response and collateral circulation compensatory capacity are important for the outcome of ischemic stroke. Here, we sought to evaluate the effect of a linarin derivate 4'-benzylapigenin-7-β-rutinoside (BLR) on neurological function and cerebral blood flow restoration in ischemic stroke. A mouse model of middle cerebral artery occlusion (30 min) with reperfusion (24 h) was used to mimic ischemic stroke in vivo, and 2,3,5-triphenyltetrazolium chloride (TTC) staining, terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assays, and immunofluorescence microscopy were used to assess the protective effects of BLR on infarct volume, neurological function, neuronal apoptosis, and inflammatory damage. Cerebral blood flow was assayed by laser speckle contrast imaging. Double immunostaining of GFAP-collagen IV and brain lucidification were performed to determine the protective effects of BLR on the disruption of brain vasculature. Differential gene expression was assessed by RNA sequencing. Coimmunoprecipitation and western blotting were used to explore the mechanism of BLR-induced neuroprotection. The results of in vivo experiments showed that BLR administration after reperfusion onset reduced infarct volume, improved neurological function, and decreased the neural cell apoptosis and inflammatory response in the ischemic brain, which was accompanied by increased cerebral blood flow and reduced detachment of astrocyte endfeet from the capillary basement membrane. The RNA sequencing data showed that BLR promoted the upregulation of extracellular matrix and angiogenesis pathway-related genes; in particular, BLR significantly increased the expression of the chondroitin sulfate proteoglycan 4 (CSPG4) gene, enhanced the membrane location of CSPG4, and promoted its downstream signaling protein expression, which is associated with KDEL receptor (KDELR) activation. In addition, activated KDELR further increased the phosphorylation of mitogen-activated protein kinases after BLR treatment. Taken together, our data showed that BLR could protect against ischemic brain injury and may serve as a new promising therapeutic candidate drug for ischemic stroke, and that KDELR might act as both a sensor and effector to activate CSPG4 to increase cerebral blood flow.
Copyright © 2022 Cong Xie et al.
Conflict of interest statement
The authors declare that they have no known conflicts of interest associated with this publication.
Figures


Similar articles
-
Beta-Boswellic Acid Protects Against Cerebral Ischemia/Reperfusion Injury via the Protein Kinase C Epsilon/Nuclear Factor Erythroid 2-like 2/Heme Oxygenase-1 Pathway.Mol Neurobiol. 2022 Jul;59(7):4242-4256. doi: 10.1007/s12035-022-02848-w. Epub 2022 May 3. Mol Neurobiol. 2022. PMID: 35505050
-
Sanpian decoction ameliorates cerebral ischemia-reperfusion injury by regulating SIRT1/ERK/HIF-1α pathway through in silico analysis and experimental validation.J Ethnopharmacol. 2024 Jan 10;318(Pt A):116898. doi: 10.1016/j.jep.2023.116898. Epub 2023 Jul 17. J Ethnopharmacol. 2024. PMID: 37467820
-
Gonadal steroids block the calpain-1-dependent intrinsic pathway of apoptosis in an experimental rat stroke model.Neurol Res. 2017 Jan;39(1):54-64. doi: 10.1080/01616412.2016.1250459. Epub 2016 Nov 11. Neurol Res. 2017. PMID: 27832728
-
Therapeutic applications of hydrogen sulfide and novel donors for cerebral ischemic stroke: a narrative review.Med Gas Res. 2023 Jan-Mar;13(1):7-9. doi: 10.4103/2045-9912.350863. Med Gas Res. 2023. PMID: 35946216 Free PMC article. Review.
-
Mechanisms Underlying Curcumin-Induced Neuroprotection in Cerebral Ischemia.Front Pharmacol. 2022 Apr 26;13:893118. doi: 10.3389/fphar.2022.893118. eCollection 2022. Front Pharmacol. 2022. PMID: 35559238 Free PMC article. Review.
Cited by
-
Abdominal Aortic Occlusion and the Inflammatory Effects in Heart and Brain.Mediators Inflamm. 2023 Nov 28;2023:2730841. doi: 10.1155/2023/2730841. eCollection 2023. Mediators Inflamm. 2023. PMID: 38131062 Free PMC article.
-
[Linarin inhibits microglia activation-mediated neuroinflammation and neuronal apoptosis in mouse spinal cord injury by inhibiting the TLR4/NF-κB pathway].Nan Fang Yi Ke Da Xue Xue Bao. 2024 Aug 20;44(8):1589-1598. doi: 10.12122/j.issn.1673-4254.2024.08.18. Nan Fang Yi Ke Da Xue Xue Bao. 2024. PMID: 39276055 Free PMC article. Chinese.
-
Mass spectrometry-based spatiotemporal characterization of lipids in early focal cerebral ischemia.Anal Bioanal Chem. 2025 Aug;417(20):4649-4662. doi: 10.1007/s00216-025-05981-2. Epub 2025 Jun 27. Anal Bioanal Chem. 2025. PMID: 40576762
-
Linarin Ameliorates Restenosis After Vascular Injury in Type 2 Diabetes Mellitus via Regulating ADAM10-Mediated Notch Signaling Pathway.Cardiovasc Toxicol. 2024 Jun;24(6):587-597. doi: 10.1007/s12012-024-09863-4. Epub 2024 May 1. Cardiovasc Toxicol. 2024. PMID: 38691303
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

