Cervical Pre-cancers: Biopsy and Immunohistochemistry
- PMID: 35928531
- PMCID: PMC9345137
- DOI: 10.25259/CMAS_03_13_2021
Cervical Pre-cancers: Biopsy and Immunohistochemistry
Abstract
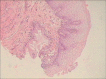
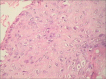
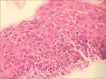
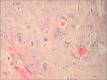
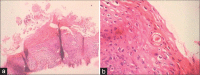
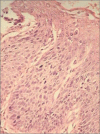
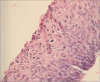
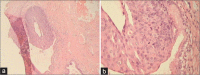
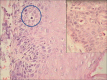
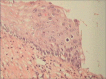
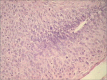
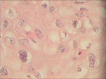
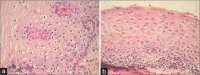
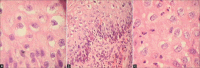
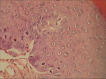
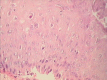
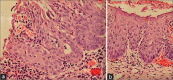
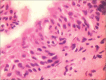
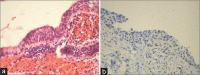
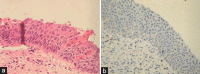
The existence of precursor lesions for invasive cervical cancer has been recognized for more than 50 years. Our understanding of the pathobiology and behavior of cervical cancer precursors has evolved considerably over the past five decades. Furthermore, the terminology used to classify pre-invasive lesions of the cervix has frequently changed. The realization that human papillomavirus (HPV) infections constitute a morphologic continuum has prompted efforts to include them within a single classification system, specifically the squamous intraepithelial lesions (SILs) which have now been embraced by the surgical pathologists. The reduced number of specific pathological categories has made clinical decision-making more straightforward. The generic criteria for SIL have two important histological parameters: Alterations in the density of superficial epithelial cells and superficial squamous atypia. The flat condyloma or cervical intraepithelial neoplasia (CIN) I is generally associated with intermediate and high-risk HPV types as against the low-risk viruses that cause exophytic/papillary growth patterns of condylomas. The diagnosis of low-grade SIL (LSIL) (flat and exophytic condylomas) requires first excluding benign mimics of LSIL and second to confirm the characteristic cytologic atypia. For high-grade SILs (HSILs), the extent and degree of atypia generally exceed the limits of that described in flat or exophytic condylomas (LSILs). Less maturation, abnormal cell differentiation, loss of cell polarity, and increased mitotic index with abnormal mitotic figures occupying increasing thickness of the epithelium define a lesion as CIN II or CIN III. Atypical immature metaplasia associated with inflammation and atrophy is a challenge in cervical biopsy interpretation. Careful attention to the growth pattern of the epithelium, the distribution of the atypia, nuclear spacing, and the degree of anisokaryosis and the presence of enlarged hyperchromatic nuclei help in differentiating a non-neoplastic from a neoplastic process. This chapter describes in depth the diagnostic difficulties in the interpretation of cervical biopsies. It also provides useful criteria in distinguishing benign mimics from true precancerous lesions and the role of biomarkers such as the p16ink4 and Ki-67 in the differential diagnosis of precursor lesions and the reactive and metaplastic epithelium.
Keywords: Atypical Immature metaplasia; Mitosis in cervical biopsy; SILs-diagnostic difficulties in cervical biopsy; biomarkers in cervical precancers; pseudokoilocytes in cervical biopsies.
© 2022 Cytopathology Foundation Inc, Published by Scientific Scholar.
Figures











Similar articles
-
Ki-67, cyclin E, and p16INK4 are complimentary surrogate biomarkers for human papilloma virus-related cervical neoplasia.Am J Surg Pathol. 2001 Jul;25(7):884-91. doi: 10.1097/00000478-200107000-00006. Am J Surg Pathol. 2001. PMID: 11420459
-
Correlation of the histologic appearance of intraepithelial neoplasia of the cervix with human papillomavirus types. Emphasis on low grade lesions including so-called flat condyloma.Int J Gynecol Pathol. 1989;8(1):18-25. doi: 10.1097/00004347-198903000-00003. Int J Gynecol Pathol. 1989. PMID: 2540101
-
P16/Ki-67 Immunostaining is Useful in Stratification of Atypical Metaplastic Epithelium of the Cervix.Clin Med Pathol. 2008;1:35-42. doi: 10.4137/cpath.s522. Epub 2008 Mar 19. Clin Med Pathol. 2008. PMID: 21876649 Free PMC article.
-
Immunohistochemistry and in situ hybridization for the diagnosis and classification of squamous lesions of the anogenital region.Semin Diagn Pathol. 2015 Sep;32(5):409-18. doi: 10.1053/j.semdp.2015.02.015. Epub 2015 Feb 7. Semin Diagn Pathol. 2015. PMID: 25862555 Review.
-
Cervical intraepithelial neoplasia.J Cell Biochem Suppl. 1995;23:71-9. doi: 10.1002/jcb.240590910. J Cell Biochem Suppl. 1995. PMID: 8747380 Review.
Cited by
-
Vaginal Adsorbent Gel as a Therapeutic Agent: Is a New Era Beginning for HPV?J Clin Med. 2025 Jul 8;14(14):4826. doi: 10.3390/jcm14144826. J Clin Med. 2025. PMID: 40725518 Free PMC article.
-
Skin Lesions Caused by HPV-A Comprehensive Review.Biomedicines. 2024 Sep 13;12(9):2098. doi: 10.3390/biomedicines12092098. Biomedicines. 2024. PMID: 39335611 Free PMC article. Review.
-
Can Mitotic Figures in Hyperchromatic Crowded Groups be Cytodiagnostic Criteria for High-Grade Squamous Intra-epithelial Lesions?J Cytol. 2024 Apr-Jun;41(2):116-122. doi: 10.4103/joc.joc_156_23. Epub 2024 Apr 24. J Cytol. 2024. PMID: 38779606 Free PMC article.
-
Human papillomavirus associated cervical lesion: pathogenesis and therapeutic interventions.MedComm (2020). 2023 Sep 14;4(5):e368. doi: 10.1002/mco2.368. eCollection 2023 Oct. MedComm (2020). 2023. PMID: 37719443 Free PMC article. Review.
-
Bridging the gap in cervical cancer screening for underserved communities: MCED and the promise of future technologies.Front Oncol. 2024 Jul 29;14:1407008. doi: 10.3389/fonc.2024.1407008. eCollection 2024. Front Oncol. 2024. PMID: 39135996 Free PMC article. Review.
References
-
- St. Clair CM, Wright JD. Cervical Intraepithelial Neoplasia: History and Detection. The Global Library of Women's Medicine's Welfare of Women. 2009 doi: 10.3843/GLOWM.10227. - DOI
-
- Lindell A. Carcinoma of the uterine cervix; incidence and influence of age; a statistical study. Acta Radiol Suppl. 1952;92:1–102. - PubMed
-
- Marisa RN, Kenneth RL, Christopher PC. In: Diagnostic Histopathology of Tumors. 4th ed. Fletcher CD, editor. Philadelphia, PA: Elsevier, Saunders; 2013. Tumors of the female genital tract cervix; pp. 814–37.
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous
