Neurological Disease-Affected Patients, including Multiple Sclerosis, Are Poor Responders to BKPyV, a Human Polyomavirus
- PMID: 35928630
- PMCID: PMC9345710
- DOI: 10.1155/2022/4864950
Neurological Disease-Affected Patients, including Multiple Sclerosis, Are Poor Responders to BKPyV, a Human Polyomavirus
Abstract
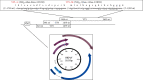
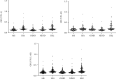
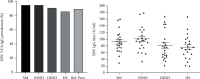
Multiple sclerosis (MS) is a neurological disease characterized by immune dysregulations. Different viruses may act as MS triggering agents. MS patients respond differently to distinct viruses. The aim of our study is to verify the association between the polyomavirus BKPyV and MS, together with other neurological diseases, through the investigation of serum IgG antibodies against the virus. Sera were from patients affected by MS and other neurologic diseases, both inflammatory (OIND) and noninflammatory (NIND). Control sera were from healthy subjects (HS). Samples were analyzed for IgG antibodies against BKPyV with an indirect ELISA with synthetic peptides mimicking the viral capsid protein 1 (VP1) antigens. As control, ELISAs were carried out to verify the immune response against the Epstein-Barr virus (EBV) of patients and controls. In addition, we assessed values for total IgG in each experimental groups. A significant lower prevalence of IgG antibodies against BKPyV VP 1 epitopes, together with a low titer, was detected in sera from MS patients and other inflammatory neurologic diseases than HS. In MS patients and OIND and NIND groups, the EBV-antibody values and total IgG did not differ from HS. Experimental data indicate that patients affected by neurological diseases, including MS, are poor responders to BKPyV VP 1 antigens, thus suggesting specific immunologic dysfunctions for this polyomavirus. Our findings are relevant in understanding the immune reactions implicated in neurological disorders.
Copyright © 2022 Ilaria Bononi et al.
Conflict of interest statement
Data of this work were enclosed, in part, in the Italian patent application number I0167478/BRE-EC/rp. In addition to this, the authors declare that there is no other conflict of interest regarding the publication of this paper.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

