Ovarian steroid hormones: A long overlooked but critical contributor to brain aging and Alzheimer's disease
- PMID: 35928995
- PMCID: PMC9344010
- DOI: 10.3389/fnagi.2022.948219
Ovarian steroid hormones: A long overlooked but critical contributor to brain aging and Alzheimer's disease
Abstract
Ovarian hormones, particularly 17β-estradiol, are involved in numerous neurophysiological and neurochemical processes, including those subserving cognitive function. Estradiol plays a key role in the neurobiology of aging, in part due to extensive interconnectivity of the neural and endocrine system. This aspect of aging is fundamental for women's brains as all women experience a drop in circulating estradiol levels in midlife, after menopause. Given the importance of estradiol for brain function, it is not surprising that up to 80% of peri-menopausal and post-menopausal women report neurological symptoms including changes in thermoregulation (vasomotor symptoms), mood, sleep, and cognitive performance. Preclinical evidence for neuroprotective effects of 17β-estradiol also indicate associations between menopause, cognitive aging, and Alzheimer's disease (AD), the most common cause of dementia affecting nearly twice more women than men. Brain imaging studies demonstrated that middle-aged women exhibit increased indicators of AD endophenotype as compared to men of the same age, with onset in perimenopause. Herein, we take a translational approach to illustrate the contribution of ovarian hormones in maintaining cognition in women, with evidence implicating menopause-related declines in 17β-estradiol in cognitive aging and AD risk. We will review research focused on the role of endogenous and exogenous estrogen exposure as a key underlying mechanism to neuropathological aging in women, with a focus on whether brain structure, function and neurochemistry respond to hormone treatment. While still in development, this research area offers a new sex-based perspective on brain aging and risk of AD, while also highlighting an urgent need for better integration between neurology, psychiatry, and women's health practices.
Keywords: Alzheimer’s disease; estrogen; hormone therapy; hormones; menopause; menstrual cycle; neuroimaging; pregnancy.
Copyright © 2022 Jett, Schelbaum, Jang, Boneu Yepez, Dyke, Pahlajani, Diaz Brinton and Mosconi.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
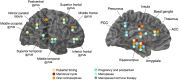
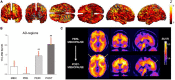
References
-
- Anderson G. L., Chlebowski R. T., Aragaki A. K., Kuller L. H., Manson J. E., Gass M., et al. (2012). Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the Women’s Health Initiative randomised placebo-controlled trial. Lancet Oncol. 13 476–486. 10.1016/S1470-2045(12)70075-X - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

