In vitro and in vivo studies on bacteria and encrustation resistance of heparin/poly-L-lysine-Cu nanoparticles coating mediated by PDA for ureteral stent application
- PMID: 35928999
- PMCID: PMC9345062
- DOI: 10.1093/rb/rbac047
In vitro and in vivo studies on bacteria and encrustation resistance of heparin/poly-L-lysine-Cu nanoparticles coating mediated by PDA for ureteral stent application
Abstract
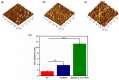
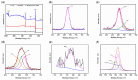
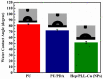
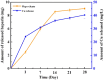
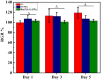
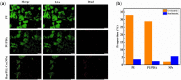
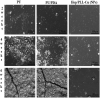
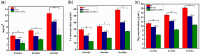
Ureteral stents are commonly utilized as a medical device to aid the flow of urine. However, biofilm formation and encrustation complications have been clinical problems. To overcome this challenge, heparin/poly-L-lysine-copper (Hep/PLL-Cu) nanoparticle was immobilized on a dopamine-coated polyurethane surface (PU/NPs). The stability and structural properties of the nanoparticles were characterized by Zeta potential, poly dispersion index, transmission electron microscopy, atom force microscopy and contact angle. The surface composition, antibacterial potency, encrustation resistance rate and biocompatibility of PU/NPs were investigated by scanning electron microscope, X-ray photoelectron spectroscopy, antibacterial assay and MTS assay, respectively. In addition, the anti-encrustation property was studied by implanting coated NPs stents in the rat bladder for 7 days. It was shown that the size and distribution of Hep/PLL-Cu nanoparticles were uniform. PU/NPs could inhibit Proteus mirabilis proliferation and biofilm formation, and exhibit no cytotoxicity. Less encrustation (Ca and Mg salt) was deposited both in vitro and in vivo on samples, demonstrating that the NPs coating could be a potential surface modification method of ureteral material for clinical use.
Keywords: biofilm; encrustation; nanoparticles; ureteral stents.
© The Author(s) 2022. Published by Oxford University Press.
Figures







Similar articles
-
In vivo study on heparin/poly-l-lysine-copper coating for surface functionalization of ureteral stent.Regen Biomater. 2022 Oct 21;9:rbac083. doi: 10.1093/rb/rbac083. eCollection 2022. Regen Biomater. 2022. PMID: 36338173 Free PMC article.
-
Polyethyleneimine brushes effectively inhibit encrustation on polyurethane ureteral stents both in dynamic bioreactor and in vivo.Mater Sci Eng C Mater Biol Appl. 2017 Feb 1;71:1166-1174. doi: 10.1016/j.msec.2016.11.125. Epub 2016 Nov 29. Mater Sci Eng C Mater Biol Appl. 2017. PMID: 27987673
-
Surface Modification of Zn-Cu Alloy with Heparin Nanoparticles for Urinary Implant Applications.ACS Appl Bio Mater. 2024 Mar 18;7(3):1748-1762. doi: 10.1021/acsabm.3c01177. Epub 2024 Mar 1. ACS Appl Bio Mater. 2024. PMID: 38428026
-
Assessment of encrustation and physicochemical properties of poly(lactide-glycolide) - Papaverine hydrochloride coating on ureteral double-J stents after long-term flow of artificial urine.J Biomed Mater Res B Appl Biomater. 2022 Feb;110(2):367-381. doi: 10.1002/jbm.b.34913. Epub 2021 Jul 24. J Biomed Mater Res B Appl Biomater. 2022. PMID: 34302425 Review.
-
Ureteral Stent Encrustation: Epidemiology, Pathophysiology, Management and Current Technology.J Urol. 2021 Jan;205(1):68-77. doi: 10.1097/JU.0000000000001343. Epub 2020 Aug 28. J Urol. 2021. PMID: 32856981 Review.
Cited by
-
New insights into the prevention of ureteral stents encrustation.Open Med (Wars). 2023 Dec 6;18(1):20230854. doi: 10.1515/med-2023-0854. eCollection 2023. Open Med (Wars). 2023. PMID: 38075034 Free PMC article. Review.
-
In vivo study on heparin/poly-l-lysine-copper coating for surface functionalization of ureteral stent.Regen Biomater. 2022 Oct 21;9:rbac083. doi: 10.1093/rb/rbac083. eCollection 2022. Regen Biomater. 2022. PMID: 36338173 Free PMC article.
-
Influence of a Composite Polylysine-Polydopamine-Quaternary Ammonium Salt Coating on Titanium on Its Ostogenic and Antibacterial Performance.Molecules. 2023 May 16;28(10):4120. doi: 10.3390/molecules28104120. Molecules. 2023. PMID: 37241863 Free PMC article.
-
Dimensional effects of surface morphology and trapped air on mammalian cell adhesion to special wetting surfaces.Regen Biomater. 2025 Apr 1;12:rbaf021. doi: 10.1093/rb/rbaf021. eCollection 2025. Regen Biomater. 2025. PMID: 40270576 Free PMC article.
References
-
- Sali GM, Joshi HB.. Ureteric stents: overview of current clinical applications and economic implications. Int J Urol 2020;27:7–15. - PubMed
-
- Donahue RP, Stamm AW, Gibbons RP, Porter CR, Kobashi KC, Corman JM, Lee UJ.. Evolution of the ureteral stent: the pivotal role of the gibbons ureteral catheter. J Urol 2018;115:3–7. - PubMed
-
- Hasirci N, Aksoy EA.. Synthesis and modifications of polyurethanes for biomedical purposes. High Perform Polym 2007;19:621–37.
-
- Wang Y, Hong Q, Chen Y, Lian X, Xiong Y.. Surface properties of polyurethanes modified by bioactive polysaccharide-based polyelectrolyte multilayers. Colloids Surf B 2012;100:77–83. - PubMed
-
- Gorman SP, Jones DS, Bonner MC, Akay M, Keane PF.. Mechanical performance of polyurethane ureteral stents in vitro and ex vivo. Biomaterials 1997;18:1379–83. - PubMed
LinkOut - more resources
Full Text Sources

