Influenza A virus elicits peri-vascular adipose tissue inflammation and vascular dysfunction of the aorta in pregnant mice
- PMID: 35930608
- PMCID: PMC9385053
- DOI: 10.1371/journal.ppat.1010703
Influenza A virus elicits peri-vascular adipose tissue inflammation and vascular dysfunction of the aorta in pregnant mice
Abstract
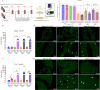
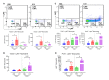
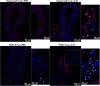
Influenza A virus (IAV) infection during pregnancy initiates significant aortic endothelial and vascular smooth muscle dysfunction, with inflammation and T cell activation, but the details of the mechanism are yet to be clearly defined. Here we demonstrate that IAV disseminates preferentially into the perivascular adipose tissue (PVAT) of the aorta in mice. IAV mRNA levels in the PVAT increased at 1-3 days post infection (d.p.i) with the levels being ~4-8 fold higher compared with the vessel wall. IAV infection also increased Ly6Clow patrolling monocytes and Ly6Chigh pro-inflammatory monocytes in the vessel wall at 3 d.p.i., which was then followed by a greater homing of these monocytes into the PVAT at 6 d.p.i. The vascular immune phenotype was characteristic of a "vascular storm"- like response, with increases in neutrophils, pro-inflammatory cytokines and oxidative stress markers in the PVAT and arterial wall, which was associated with an impairment in endothelium-dependent relaxation to acetylcholine. IAV also triggered a PVAT compartmentalised elevation in CD4+ and CD8+ activated T cells. In conclusion, the PVAT of the aorta is a niche that supports IAV dissemination and a site for perpetuating a profound innate inflammatory and adaptive T cell response. The manifestation of this inflammatory response in the PVAT following IAV infection may be central to the genesis of cardiovascular complications arising during pregnancy.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
Gestational influenza A virus infection elicits nonresolving vascular dysfunction and T-cell accumulation in the aorta of mice.Am J Physiol Heart Circ Physiol. 2024 Oct 1;327(4):H967-H977. doi: 10.1152/ajpheart.00646.2023. Epub 2024 Sep 6. Am J Physiol Heart Circ Physiol. 2024. PMID: 39240256
-
Influenza A virus causes maternal and fetal pathology via innate and adaptive vascular inflammation in mice.Proc Natl Acad Sci U S A. 2020 Oct 6;117(40):24964-24973. doi: 10.1073/pnas.2006905117. Epub 2020 Sep 21. Proc Natl Acad Sci U S A. 2020. PMID: 32958663 Free PMC article.
-
Gestational intermittent hypoxia induces endothelial dysfunction, reduces perivascular adiponectin and causes epigenetic changes in adult male offspring.J Physiol. 2019 Nov;597(22):5349-5364. doi: 10.1113/JP277936. Epub 2019 Aug 22. J Physiol. 2019. PMID: 31441069
-
The Role of Obesity-Induced Perivascular Adipose Tissue (PVAT) Dysfunction in Vascular Homeostasis.Nutrients. 2021 Oct 28;13(11):3843. doi: 10.3390/nu13113843. Nutrients. 2021. PMID: 34836100 Free PMC article. Review.
-
Perivascular adipose tissue in age-related vascular disease.Ageing Res Rev. 2020 May;59:101040. doi: 10.1016/j.arr.2020.101040. Epub 2020 Feb 26. Ageing Res Rev. 2020. PMID: 32112889 Review.
Cited by
-
Gestational influenza A virus infection elicits nonresolving vascular dysfunction and T-cell accumulation in the aorta of mice.Am J Physiol Heart Circ Physiol. 2024 Oct 1;327(4):H967-H977. doi: 10.1152/ajpheart.00646.2023. Epub 2024 Sep 6. Am J Physiol Heart Circ Physiol. 2024. PMID: 39240256
-
Biological Sex and Pregnancy Affect Influenza Pathogenesis and Vaccination.Curr Top Microbiol Immunol. 2023;441:111-137. doi: 10.1007/978-3-031-35139-6_5. Curr Top Microbiol Immunol. 2023. PMID: 37695427
-
Influenza Virus Infection during Pregnancy as a Trigger of Acute and Chronic Complications.Viruses. 2022 Dec 7;14(12):2729. doi: 10.3390/v14122729. Viruses. 2022. PMID: 36560733 Free PMC article. Review.
-
Beyond energy balance regulation: The underestimated role of adipose tissues in host defense against pathogens.Front Immunol. 2023 Mar 2;14:1083191. doi: 10.3389/fimmu.2023.1083191. eCollection 2023. Front Immunol. 2023. PMID: 36936928 Free PMC article. Review.
-
Understanding Comorbidities of Respiratory Models as Novel Platforms for Drug Discovery.ACS Pharmacol Transl Sci. 2024 Oct 25;7(11):3385-3393. doi: 10.1021/acsptsci.4c00484. eCollection 2024 Nov 8. ACS Pharmacol Transl Sci. 2024. PMID: 39539266 Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

