Stem cell-based therapy for human diseases
- PMID: 35933430
- PMCID: PMC9357075
- DOI: 10.1038/s41392-022-01134-4
Stem cell-based therapy for human diseases
Abstract
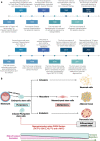
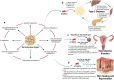
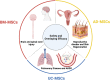
Recent advancements in stem cell technology open a new door for patients suffering from diseases and disorders that have yet to be treated. Stem cell-based therapy, including human pluripotent stem cells (hPSCs) and multipotent mesenchymal stem cells (MSCs), has recently emerged as a key player in regenerative medicine. hPSCs are defined as self-renewable cell types conferring the ability to differentiate into various cellular phenotypes of the human body, including three germ layers. MSCs are multipotent progenitor cells possessing self-renewal ability (limited in vitro) and differentiation potential into mesenchymal lineages, according to the International Society for Cell and Gene Therapy (ISCT). This review provides an update on recent clinical applications using either hPSCs or MSCs derived from bone marrow (BM), adipose tissue (AT), or the umbilical cord (UC) for the treatment of human diseases, including neurological disorders, pulmonary dysfunctions, metabolic/endocrine-related diseases, reproductive disorders, skin burns, and cardiovascular conditions. Moreover, we discuss our own clinical trial experiences on targeted therapies using MSCs in a clinical setting, and we propose and discuss the MSC tissue origin concept and how MSC origin may contribute to the role of MSCs in downstream applications, with the ultimate objective of facilitating translational research in regenerative medicine into clinical applications. The mechanisms discussed here support the proposed hypothesis that BM-MSCs are potentially good candidates for brain and spinal cord injury treatment, AT-MSCs are potentially good candidates for reproductive disorder treatment and skin regeneration, and UC-MSCs are potentially good candidates for pulmonary disease and acute respiratory distress syndrome treatment.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

