Fetal Brain Damage in Human Fetuses with Congenital Cytomegalovirus Infection: Histological Features and Viral Tropism
- PMID: 35933637
- PMCID: PMC10006254
- DOI: 10.1007/s10571-022-01258-9
Fetal Brain Damage in Human Fetuses with Congenital Cytomegalovirus Infection: Histological Features and Viral Tropism
Abstract
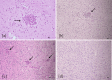
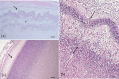
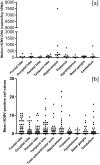
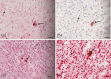
Human cytomegalovirus (HCMV) causes congenital neurological lifelong disabilities. To date, the neuropathogenesis of brain injury related to congenital HCMV (cCMV) infection is poorly understood. This study evaluates the characteristics and pathogenetic mechanisms of encephalic damage in cCMV infection. Ten HCMV-infected human fetuses at 21 weeks of gestation were examined. Specifically, tissues from different brain areas were analyzed by: (i) immunohistochemistry (IHC) to detect HCMV-infected cell distribution, (ii) hematoxylin-eosin staining to evaluate histological damage and (iii) real-time PCR to quantify tissue viral load (HCMV-DNA). The differentiation stage of HCMV-infected neural/neuronal cells was assessed by double IHC to detect simultaneously HCMV-antigens and neural/neuronal markers: nestin (a marker of neural stem/progenitor cells), doublecortin (DCX, marker of cells committed to the neuronal lineage) and neuronal nuclei (NeuN, identifying mature neurons). HCMV-positive cells and viral DNA were found in the brain of 8/10 (80%) fetuses. For these cases, brain damage was classified as mild (n = 4, 50%), moderate (n = 3, 37.5%) and severe (n = 1, 12.5%) based on presence and frequency of pathological findings (necrosis, microglial nodules, microglial activation, astrocytosis, and vascular changes). The highest median HCMV-DNA level was found in the hippocampus (212 copies/5 ng of human DNA [hDNA], range: 10-7,505) as well as the highest mean HCMV-infected cell value (2.9 cells, range: 0-23), followed by that detected in subventricular zone (1.7 cells, range: 0-19). These findings suggested a preferential viral tropism for both neural stem/progenitor cells and neuronal committed cells, residing in these regions, confirmed by the expression of DCX and nestin in 94% and 63.3% of HCMV-positive cells, respectively. NeuN was not found among HCMV-positive cells and was nearly absent in the brain with severe damage, suggesting HCMV does not infect mature neurons and immature neural/neuronal cells do not differentiate into neurons. This could lead to known structural and functional brain defects from cCMV infection.
Keywords: Brain damage; Congenital infection; Cytomegalovirus; Human fetuses; Neural/neuronal cells; Viral tropism.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no conflicts of interest/competing interests.
Figures



Similar articles
-
Nitric Oxide Attenuates Human Cytomegalovirus Infection yet Disrupts Neural Cell Differentiation and Tissue Organization.J Virol. 2022 Jul 27;96(14):e0012622. doi: 10.1128/jvi.00126-22. Epub 2022 Jul 7. J Virol. 2022. PMID: 35862705 Free PMC article.
-
Human neural progenitor cell models to study the antiviral effects and neuroprotective potential of approved and investigational human cytomegalovirus inhibitors.Antiviral Res. 2024 Mar;223:105816. doi: 10.1016/j.antiviral.2024.105816. Epub 2024 Jan 28. Antiviral Res. 2024. PMID: 38286212
-
Human Cytomegalovirus Compromises Development of Cerebral Organoids.J Virol. 2019 Aug 13;93(17):e00957-19. doi: 10.1128/JVI.00957-19. Print 2019 Sep 1. J Virol. 2019. PMID: 31217239 Free PMC article.
-
Diagnosis and prognostic markers of HCMV infection.J Clin Virol. 2001 Jun;21(3):213-21. doi: 10.1016/s1386-6532(00)00164-5. J Clin Virol. 2001. PMID: 11397657 Review.
-
Overview of Human Cytomegalovirus Pathogenesis.Methods Mol Biol. 2021;2244:1-18. doi: 10.1007/978-1-0716-1111-1_1. Methods Mol Biol. 2021. PMID: 33555579 Review.
Cited by
-
Stabilizing microtubules aids neurite structure and disrupts syncytia formation in human cytomegalovirus-infected human forebrain neurons.bioRxiv [Preprint]. 2024 Aug 19:2024.08.16.608340. doi: 10.1101/2024.08.16.608340. bioRxiv. 2024. PMID: 39229072 Free PMC article. Preprint.
-
Human cytomegalovirus infection impairs neural differentiation via repressing sterol regulatory element binding protein 2-mediated cholesterol biosynthesis.Cell Mol Life Sci. 2024 Jul 6;81(1):289. doi: 10.1007/s00018-024-05278-0. Cell Mol Life Sci. 2024. PMID: 38970696 Free PMC article.
-
Neuron-restricted cytomegalovirus latency in the central nervous system regulated by CD4+ T-cells and IFN-γ.J Neuroinflammation. 2025 Mar 29;22(1):95. doi: 10.1186/s12974-025-03422-6. J Neuroinflammation. 2025. PMID: 40158177 Free PMC article.
-
Human Cytomegalovirus Infection and Neurocognitive and Neuropsychiatric Health.Pathogens. 2024 May 16;13(5):417. doi: 10.3390/pathogens13050417. Pathogens. 2024. PMID: 38787269 Free PMC article. Review.
-
HCMV Infection Reduces Nidogen-1 Expression, Contributing to Impaired Neural Rosette Development in Brain Organoids.J Virol. 2023 May 31;97(5):e0171822. doi: 10.1128/jvi.01718-22. Epub 2023 May 1. J Virol. 2023. PMID: 37125912 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

