Recent advances in PLGA micro/nanoparticle delivery systems as novel therapeutic approach for drug-resistant tuberculosis
- PMID: 35935487
- PMCID: PMC9355142
- DOI: 10.3389/fbioe.2022.941077
Recent advances in PLGA micro/nanoparticle delivery systems as novel therapeutic approach for drug-resistant tuberculosis
Abstract
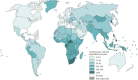
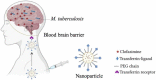
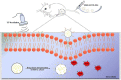
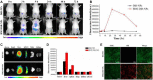
Tuberculosis is a severe infectious disease caused by Mycobacterium tuberculosis and is a significant public health concern globally. The World Health Organization (WHO) recommends a combination regimen of several drugs, such as rifampicin (RIF), isoniazid (INH), pyrazinamide (PZA), and ethambutol (ETB), to treat tuberculosis. However, these drugs have low plasma concentrations after oral administration and require multiple high doses, which may lead to the occurrence and development of drug-resistant tuberculosis. Micro/Nanotechnology drug delivery systems have considerable potential in treating drug-resistant tuberculosis, allowing the sustained release of the drug and delivery of the drug to a specific target. These system properties could improve drug bioavailability, reduce the dose and frequency of administration, and solve the problem of non-adherence to the prescribed therapy. This study systematically reviewed the recent advances in PLGA micro/nanoparticle delivery systems as a novel therapeutic approach for drug-resistant tuberculosis.
Keywords: Mycobacterium tuberculosis; PLGA microparticles; PLGA nanoparticles; combination therapy; drug-resistant tuberculosis; inhalable therapy.
Copyright © 2022 Shao, Shen and Liu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures







References
-
- Abdelghany S., Parumasivam T., Pang A., Roediger B., Tang P., Jahn K., et al. (2019). Alginate modified-PLGA nanoparticles entrapping amikacin and moxifloxacin as a novel host-directed therapy for multidrug-resistant tuberculosis. J. Drug Deliv. Sci. Technol. 52, 642–651. 10.1016/j.jddst.2019.05.025 - DOI
-
- Al-Qushawi A., Rassouli A., Atyabi F., Peighambari S. M., Esfandyari-Manesh M., Shams G. R., et al. (2016). Preparation and characterization of three tilmicosin-loaded lipid nanoparticles: Physicochemical properties and in-vitro antibacterial activities. Iran. J. Pharm. Res. 15 (4), 663–676. - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

