Intratumoral IL-28B Gene Delivery Elicits Antitumor Effects by Remodeling of the Tumor Microenvironment in H22-Bearing Mice
- PMID: 35935577
- PMCID: PMC9352479
- DOI: 10.1155/2022/1345971
Intratumoral IL-28B Gene Delivery Elicits Antitumor Effects by Remodeling of the Tumor Microenvironment in H22-Bearing Mice
Abstract
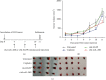
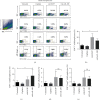
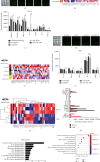
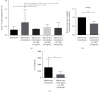
IL-28B, belonging to type III interferons (IFN-λs), exhibits a potent antitumor activity with reduced regulated T cells (Tregs) population, yet the effect of IL-28B on the tumor microenvironment (TME) and if IL-28B can downregulate Tregs directly in vitro are still unknown. In this study, we investigated the effects of IL-28B on Tregs in the spleen and TME in H22 tumor-bearing mice and verified the downregulation of IL-28B on Tregs in vitro. We found that rAd-mIL-28B significantly inhibited tumor growth and reduced the frequency of splenic CD4+Foxp3+ T cells. The levels of CXCL13, ICAM-1, MCP-5, and IL-7 in the serum, and the levels of IL-15 and sFasL in the tumor tissue decreased significantly after rAd-mIL-28B treatment relative to rAd-EGFP. Furthermore, the percentage of CD8+ cells in the TME was significantly increased in the rAd-mIL-28B group compared with the untreated group. In vitro, splenocytes were stimulated with anti-CD3/CD28 and IL-2 in the presence of TGF-β with or without IL-28B for three days and followed by flow cytometric, RT-PCR, and IL-10 production analysis. The results showed that IL-28B significantly reduced the proportion of induced Foxp3+ cells. It demonstrated that IL-28B may be used as a promising immunotherapy strategy against cancer.
Copyright © 2022 Zhi Li et al.
Conflict of interest statement
The authors declare no potential conflicts of interest.
Figures

References
-
- Nardone V., Botta C., Caraglia M., et al. Tumor infiltrating T lymphocytes expressing FoxP3, CCR7 or PD-1 predict the outcome of prostate cancer patients subjected to salvage radiotherapy after biochemical relapse. Cancer Biology & Therapy . 2016;17(11):1213–1220. doi: 10.1080/15384047.2016.1235666. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

