Monocyte Metabolism and Function in Patients Undergoing Cardiac Surgery
- PMID: 35935635
- PMCID: PMC9347004
- DOI: 10.3389/fcvm.2022.853967
Monocyte Metabolism and Function in Patients Undergoing Cardiac Surgery
Abstract
Objective: Cardiopulmonary bypass (CPB) can lead to systemic inflammation, which is associated with higher morbidity. Therefore, we investigated the metabolism of isolated blood monocytes before and after CPB compared to healthy controls.
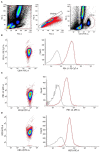
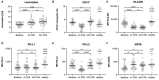
Methods: In this prospective, monocentric, observational study, we included 30 patients undergoing CPB and 20 controls. We isolated monocytes from heparinized blood and investigated their metabolism by using Seahorse technology before (t0), 4 h (t4), and 24 h (t24) after the start of the CPB. We also examined programmed cell death 1 ligand (PD-L1), PD-L2, V-domain Ig suppressor of T cell activation (VISTA), and human leukocyte antigen-DR isotype (HLA-DR) using fluorescence-activated cell sorting analysis. Additionally, we investigated plasma cytokine levels in patients without and after ex vivo stimulation.
Results: CPB-induced inflammatory responses are shown by significantly elevated plasma interleukin-6 levels in the CPB group compared to baseline and controls [t0: 0 ng/ml (95%CI 0-0 ng/ml); t4: 0.16 ng/ml (95%CI 0.1-0.197 ng/ml), p < 0.0001; t24: 0.11 ng/ml (95% CI 0.1-0.16 ng/ml), p < 0.0001, and controls: 0 ng/ml (95% CI 0-0 ng/ml)]. The cytokine release in the ex vivo stimulation is reduced for lipopolysaccharide stimulation at t4 [t0: 35.68 ng/ml (95% CI 22.17-46.57 ng/ml) vs. t4: 15.02 (95% CI 10.25-24.78 ng/ml), p < 0.0001]. Intracellular metabolism of monocytes after CPB showed a protracted shift to aerobic glycolysis [t0: 179.2 pmol/min (95% CI 138.0-205.1 pmol/min) vs. t24: 250.1 pmol/min (95% CI 94.8-300.2 pmol/min), p < 0.0001]. Additionally, we observed an altered metabolism in monocytes in patients undergoing cardiac surgery compared to controls even before any surgical procedure [t0: 179.2 pmol/min (95% CI 138.0-205.1) vs. controls 97.4 (95% CI 59.13-144.6 pmol/min), p = 0.0031].
Conclusion: After CPB, patients' monocytes show a shift in metabolism from oxidative phosphorylation to aerobic glycolysis, which is associated with energy-demanding and proinflammatory processes. This is the first study to show changes in monocyte immunometabolism in cardiac surgery. Monocytes of patients undergoing cardiac surgery were leaning toward aerobic glycolysis even before any surgical procedure was conducted. Leaving the question of the pathophysiological mechanisms for future studies to be investigated and paving the way for potential therapy approaches preventing inflammatory effects of CPB.
Keywords: Warburg effect; cardiac surgery; immune reaction; immunometabolism; inflammation; monocytes.
Copyright © 2022 Mayer, Altvater, Schenz, Arif, Karck, Leuschner, Weigand, Uhle and Lichtenstern.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Granulocyte-macrophage colony-stimulating factor (GM-CSF) restores decreased monocyte HLA-DR expression after cardiopulmonary bypass.Thorac Cardiovasc Surg. 2007 Feb;55(1):24-31. doi: 10.1055/s-2006-924621. Thorac Cardiovasc Surg. 2007. PMID: 17285470
-
Monitoring of monocyte functional state after extracorporeal circulation: a flow cytometry study.Cytometry B Clin Cytom. 2004 Mar;58(1):17-24. doi: 10.1002/cyto.b.10061. Cytometry B Clin Cytom. 2004. PMID: 14994371
-
Immunodepression after CPB: Cytokine dynamics and clinics after pediatric cardiac surgery - A prospective trial.Cytokine. 2019 Oct;122:154018. doi: 10.1016/j.cyto.2017.03.017. Epub 2017 Apr 12. Cytokine. 2019. PMID: 28411047 Clinical Trial.
-
Pediatric cardiac surgery with cardiopulmonary bypass: pathways contributing to transient systemic immune suppression.Shock. 2001;16 Suppl 1:24-32. doi: 10.1097/00024382-200116001-00006. Shock. 2001. PMID: 11770029 Review.
-
Amplification of the inflammatory response: adhesion molecules associated with platelet/white cell responses.J Cardiovasc Pharmacol. 1996;27 Suppl 1:S6-12. doi: 10.1097/00005344-199600001-00005. J Cardiovasc Pharmacol. 1996. PMID: 8938278 Review.
Cited by
-
Metabolism Serves as a Bridge Between Cardiomyocytes and Immune Cells in Cardiovascular Diseases.Cardiovasc Drugs Ther. 2025 Jun;39(3):661-676. doi: 10.1007/s10557-024-07545-5. Epub 2024 Jan 18. Cardiovasc Drugs Ther. 2025. PMID: 38236378 Review.
-
Early dynamic changes to monocytes following major surgery are associated with subsequent infections.Front Immunol. 2024 Apr 9;15:1352556. doi: 10.3389/fimmu.2024.1352556. eCollection 2024. Front Immunol. 2024. PMID: 38655251 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials

