Factors associated with mood disorders and the efficacy of the targeted treatment of functional dyspepsia: A randomized clinical trial
- PMID: 35935797
- PMCID: PMC9353185
- DOI: 10.3389/fmed.2022.859661
Factors associated with mood disorders and the efficacy of the targeted treatment of functional dyspepsia: A randomized clinical trial
Abstract
Background: Patients with functional dyspepsia (FD) are often accompanied by mood disorders (MDs). This study aimed to identify factors associated with MDs in patients with FD and evaluate the efficacy of targeted treatment plans.
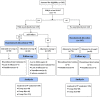
Methods: Relevant scales were used to assess MDs. Patients with FD having MDs and acid reflux were treated with flupentixol and melitracen (FM) and acid-suppressive therapy (AST) (histamine-2 receptor antagonists (H2RAs) (group A) or proton pump inhibitors (PPIs) (group B)), and those without acid reflux (group C) did not receive AST. Patients with FD without MDs were randomly administered H2RAs (group D) or PPIs (group E). The primary endpoints were factors associated with MDs and improvement in gastrointestinal (GI) symptoms and MDs in patients with FD.
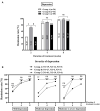
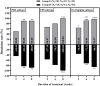
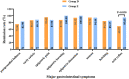
Results: A total of 362 patients with FD were enrolled in this study. Patients with FD having high GI score and low education were found prone to MDs. At week 2, the remission rate of overall GI symptoms and depression was significantly higher in group B than that in groups A and C [GI: 72.72% (32/44) vs. 47.73% (21/44) and 72.72% (32/44) vs. 38.94% (44/113), all P < 0.05; depression: 72.22% (26/36) vs. 41.67% (15/36) and 72.22% (26/36) vs. 41.57% (37/89), all P < 0.05]. Furthermore, the remission rate of overall GI symptoms was significantly higher in group E than that in group D [60.29% (41/68) vs. 42.65% (29/68), P < 0.05]. At week 8, similar efficacies and adverse reactions were observed in these groups.
Conclusion: The risk factors for MDs were high GI scores and low literacy rates. Thus, targeted treatment (FM+PPIs for patients with MDs; PPIs for patients without MDs) can improve the efficacy of patients with FD.
Clinical trial registration: www.chictr.org.cn, identifier ChiCTR2100053126.
Keywords: H2RAs; PPIs; dyspepsia; efficacy; psychotropic medications.
Copyright © 2022 Huang, Zheng, Cai, Zhang, Su and Wang.
Figures

Similar articles
-
Effect of clinician-patient communication on compliance with flupentixol-melitracen in functional dyspepsia patients.World J Gastroenterol. 2015 Apr 21;21(15):4652-9. doi: 10.3748/wjg.v21.i15.4652. World J Gastroenterol. 2015. PMID: 25914475 Free PMC article. Clinical Trial.
-
Randomized clinical trial: the effects of mirtazapine in functional dyspepsia patients.Therap Adv Gastroenterol. 2025 Jan 3;18:17562848241311129. doi: 10.1177/17562848241311129. eCollection 2025. Therap Adv Gastroenterol. 2025. PMID: 39758962 Free PMC article.
-
Chinese Herbal Medicine for Functional Dyspepsia With Psychological Disorders: A Systematic Review and Meta-Analysis.Front Neurosci. 2022 Jul 14;16:933290. doi: 10.3389/fnins.2022.933290. eCollection 2022. Front Neurosci. 2022. PMID: 35911981 Free PMC article.
-
Impact of sleep disorders in Japanese patients with functional dyspepsia (FD): nizatidine improves clinical symptoms, gastric emptying and sleep disorders in FD patients.J Gastroenterol Hepatol. 2013 Aug;28(8):1314-20. doi: 10.1111/jgh.12236. J Gastroenterol Hepatol. 2013. PMID: 23611167 Clinical Trial.
-
Insights into the evaluation and management of dyspepsia: recent developments and new guidelines.Therap Adv Gastroenterol. 2018 Oct 30;11:1756284818805597. doi: 10.1177/1756284818805597. eCollection 2018. Therap Adv Gastroenterol. 2018. PMID: 30397412 Free PMC article. Review.
Cited by
-
Global trends in research related to functional dyspepsia and anxiety or depression over the past two decades: a bibliometric analysis.Front Neurosci. 2023 Nov 2;17:1218001. doi: 10.3389/fnins.2023.1218001. eCollection 2023. Front Neurosci. 2023. PMID: 38027507 Free PMC article.
-
Flupentixol/melitracen for epigastric pain syndrome in female patients with abnormal urinalysis and therapeutic insights into the brain-bladder axis.Sci Rep. 2025 Jul 13;15(1):25331. doi: 10.1038/s41598-025-10749-w. Sci Rep. 2025. PMID: 40653504 Free PMC article. Clinical Trial.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

