Population pharmacokinetics and initial dose optimization of tacrolimus in children with severe combined immunodeficiency undergoing hematopoietic stem cell transplantation
- PMID: 35935844
- PMCID: PMC9354257
- DOI: 10.3389/fphar.2022.869939
Population pharmacokinetics and initial dose optimization of tacrolimus in children with severe combined immunodeficiency undergoing hematopoietic stem cell transplantation
Abstract
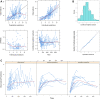
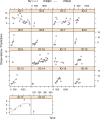
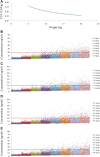
The present study aimed to explore the population pharmacokinetics and initial dose optimization of tacrolimus in children with severe combined immunodeficiency (SCID) undergoing hematopoietic stem cell transplantation (HSCT). Children with SCID undergoing HSCT treated with tacrolimus were enrolled for analysis. Population pharmacokinetics of tacrolimus was built up by a nonlinear mixed-effects model (NONMEM), and initial dose optimization of tacrolimus was simulated with the Monte Carlo method in children weighing <20 kg at different doses. A total of 18 children with SCID undergoing HSCT were included for analysis, with 130 tacrolimus concentrations. Body weight was included as a covariable in the final model. Tacrolimus CL/F was 0.36-0.26 L/h/kg from body weights of 5-20 kg. Meanwhile, we simulated the tacrolimus concentrations using different body weights (5-20 kg) and different dose regimens (0.1-0.8 mg/kg/day). Finally, the initial dose regimen of 0.6 mg/kg/day tacrolimus was recommended for children with SCID undergoing HSCT whose body weights were 5-20 kg. It was the first time to establish tacrolimus population pharmacokinetics in children with SCID undergoing HSCT; in addition, the initial dose optimization of tacrolimus was recommended.
Keywords: hematopoietic stem cell transplantation; initial dose optimization; population pharmacokinetics; severe combined immunodeficiency; tacrolimus.
Copyright © 2022 Chen, Wang, Zheng, Zhai, Xu and Li.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Cojutti P. G., Morandin E., Baraldo M., Pea F. (2021). Population pharmacokinetics of continuous infusion of piperacillin/tazobactam in very elderly hospitalized patients and considerations for target attainment against Enterobacterales and Pseudomonas aeruginosa . Int. J. Antimicrob. Agents 58 (4), 106408. 10.1016/j.ijantimicag.2021.106408 - DOI - PubMed
LinkOut - more resources
Full Text Sources

