How to Achieve Sufficient Endogenous Insulin Suppression in Euglycemic Clamps Assessing the Pharmacokinetics and Pharmacodynamics of Long-Acting Insulin Preparations Employing Healthy Volunteers
- PMID: 35935883
- PMCID: PMC9354408
- DOI: 10.3389/fphar.2022.899798
How to Achieve Sufficient Endogenous Insulin Suppression in Euglycemic Clamps Assessing the Pharmacokinetics and Pharmacodynamics of Long-Acting Insulin Preparations Employing Healthy Volunteers
Abstract
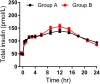
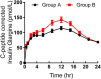
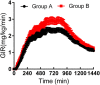
The therapeutic effect of basal insulin analogs will be sustained at a rather low insulin level. When employing healthy volunteers to assess the pharmacokinetics (PK) and pharmacodynamics (PD) of long-acting insulin preparations by euglycemic clamp techniques, endogenous insulin cannot be ignored and sufficient endogenous insulin inhibition is crucial for the PD and/or PK assessment. This study aimed to explore a way to sufficiently inhibit endogenous insulin secretion. Healthy Chinese male and female volunteers were enrolled. After a subcutaneous injection of insulin glargine (IGlar) (LY2963016 or Lantus) (0.5 IU/kg), they underwent a manual euglycemic clamp for up to 24 h where the target blood glucose (BG) was set as 0.28 mmol/L below the individual's baseline. Blood samples were collected for analysis of PK/PD and C-peptide. The subjects fell into two groups according to the reduction extent of postdose C-peptide from baseline. After matching for the dosage proportion of Lantus, there were 52 subjects in group A (C-peptide reduction<50%) and 26 in group B (C-peptide reduction≥50%), respectively. No significant difference was detected in age, body mass index, the proportion of Latus treatment and female participants. A lower basal BG was observed in group B compared to group A (4.35 ± 0.26 vs. 4.59 ± 0.22 mmol/L, p < 0.05). The clamp studies were all conducted with high quality (where BG was consistently maintained around the target and exhibited a low variety). The binary logistic regression analysis indicated low basal BG as an independent factor for the success of sufficient endogenous insulin suppression. In conclusion, setting a lower sub-baseline target BG (e.g., 10% instead of 5% below baseline) might be an approach to help achieve sufficient endogenous insulin suppression in euglycemic clamps with higher basal BG levels (e.g., beyond 4.60 mmol/L).
Keywords: endogenous insulin suppression; euglycemic clamp; healthy volunteers; pharmacodynamics; pharmacokinetics.
Copyright © 2022 Liu, Li, Yu, Li, Tan and Yu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
LinkOut - more resources
Full Text Sources

