Immunogenicity of a vaccinia virus-based severe acute respiratory syndrome coronavirus 2 vaccine candidate
- PMID: 35935962
- PMCID: PMC9353262
- DOI: 10.3389/fimmu.2022.911164
Immunogenicity of a vaccinia virus-based severe acute respiratory syndrome coronavirus 2 vaccine candidate
Abstract
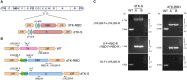
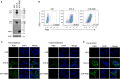
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccines provide essential tools for the control of the COVID-19 pandemic. A number of technologies have been employed to develop SARS-CoV-2 vaccines, including the inactivated SARS-CoV-2 particles, mRNA to express viral spike protein, recombinant spike proteins, and viral vectors. Here, we report the use of the vaccinia virus Tiantan strain as a vector to express the SARS-CoV-2 spike protein. When it was used to inoculate mice, robust SARS-CoV-2 spike protein-specific antibody response and T-cell response were detected. Sera from the vaccinated mice showed strong neutralizing activity against the ancestral Wuhan SARS-CoV-2, the variants of concern (VOCs) B.1.351, B.1.617.2, and the emerging B.1.1.529 (omicron). This finding supports the possibility of developing a new type of SARS-CoV-2 vaccine using the vaccinia virus vector.
Keywords: COVID-19; SARS-CoV-2; spike protein; vaccine; vaccinia virus.
Copyright © 2022 Mei, Fan, Liu, Zhao, Huang, Wei, Hu, Xie, Wang, Ai, Liang, Xu and Guo.
Conflict of interest statement
FG, FX, SM, and ZF are inventors on pending and issued patents on SARS-CoV-2 vaccines. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- World Health Organization . Who coronavirus (Covid-19) dashboard. Available at: https://covid19.who.int/.
-
- Zhu FC, Li YH, Guan XH, Hou LH, Wang WJ, Li JX, et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored covid-19 vaccine: A dose-escalation, open-label, non-randomised, first-in-Human trial. Lancet (2020) 395(10240):1845–54. doi: 10.1016/S0140-6736(20)31208-3 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

