Efficient cellular and humoral immune response and production of virus-neutralizing antibodies by the Hepatitis B Virus S/preS116-42 antigen
- PMID: 35935966
- PMCID: PMC9354405
- DOI: 10.3389/fimmu.2022.941243
Efficient cellular and humoral immune response and production of virus-neutralizing antibodies by the Hepatitis B Virus S/preS116-42 antigen
Abstract
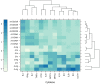
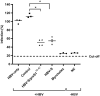
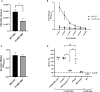
Despite the availability of improved antiviral therapies, infection with Hepatitis B virus (HBV) remains a3 significant health issue, as a curable treatment is yet to be discovered. Current HBV vaccines relaying on the efficient expression of the small (S) envelope protein in yeast and the implementation of mass vaccination programs have clearly contributed to containment of the disease. However, the lack of an efficient immune response in up to 10% of vaccinated adults, the controversies regarding the seroprotection persistence in vaccine responders and the emergence of vaccine escape virus mutations urge for the development of better HBV immunogens. Due to the critical role played by the preS1 domain of the large (L) envelope protein in HBV infection and its ability to trigger virus neutralizing antibodies, including this protein in novel vaccine formulations has been considered a promising strategy to overcome the limitations of S only-based vaccines. In this work we aimed to combine relevant L and S epitopes in chimeric antigens, by inserting preS1 sequences within the external antigenic loop of S, followed by production in mammalian cells and detailed analysis of their antigenic and immunogenic properties. Of the newly designed antigens, the S/preS116-42 protein assembled in subviral particles (SVP) showed the highest expression and secretion levels, therefore, it was selected for further studies in vivo. Analysis of the immune response induced in mice vaccinated with S/preS116-42- and S-SVPs, respectively, demonstrated enhanced immunogenicity of the former and its ability to activate both humoral and cellular immune responses. This combined activation resulted in production of neutralizing antibodies against both wild-type and vaccine-escape HBV variants. Our results validate the design of chimeric HBV antigens and promote the novel S/preS1 protein as a potential vaccine candidate for administration in poor-responders to current HBV vaccines.
Keywords: HBV; SVP; adjuvants; antigens; chimeric proteins; vaccine design.
Copyright © 2022 Pantazica, Dobrica, Lazar, Scurtu, Tucureanu, Caras, Ionescu, Costache, Onu, Clarke, Stavaru and Branza-Nichita.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
A novel chimeric Hepatitis B virus S/preS1 antigen produced in mammalian and plant cells elicits stronger humoral and cellular immune response than the standard vaccine-constituent, S protein.Antiviral Res. 2017 Aug;144:256-265. doi: 10.1016/j.antiviral.2017.06.017. Epub 2017 Jun 27. Antiviral Res. 2017. PMID: 28666757
-
Oral administration of a chimeric Hepatitis B Virus S/preS1 antigen produced in lettuce triggers infection neutralizing antibodies in mice.Vaccine. 2018 Sep 11;36(38):5789-5795. doi: 10.1016/j.vaccine.2018.07.072. Epub 2018 Aug 3. Vaccine. 2018. PMID: 30082163
-
The "humanized" N-glycosylation pathway in CRISPR/Cas9-edited Nicotiana benthamiana significantly enhances the immunogenicity of a S/preS1 Hepatitis B Virus antigen and the virus-neutralizing antibody response in vaccinated mice.Plant Biotechnol J. 2023 Jun;21(6):1176-1190. doi: 10.1111/pbi.14028. Epub 2023 Mar 10. Plant Biotechnol J. 2023. PMID: 36779605 Free PMC article.
-
Applications of human hepatitis B virus preS domain in bio- and nanotechnology.World J Gastroenterol. 2015 Jun 28;21(24):7400-11. doi: 10.3748/wjg.v21.i24.7400. World J Gastroenterol. 2015. PMID: 26139986 Free PMC article. Review.
-
Hepatitis B Virus (HBV) Subviral Particles as Protective Vaccines and Vaccine Platforms.Viruses. 2020 Jan 21;12(2):126. doi: 10.3390/v12020126. Viruses. 2020. PMID: 31973017 Free PMC article. Review.
Cited by
-
Plant-made vaccines against viral diseases in humans and farm animals.Front Plant Sci. 2023 Mar 28;14:1170815. doi: 10.3389/fpls.2023.1170815. eCollection 2023. Front Plant Sci. 2023. PMID: 37056490 Free PMC article. Review.
-
Hepatitis B and D virus entry.Nat Rev Microbiol. 2025 May;23(5):318-331. doi: 10.1038/s41579-024-01121-2. Epub 2024 Nov 21. Nat Rev Microbiol. 2025. PMID: 39572840 Review.
-
Prokaryote- and Eukaryote-Based Expression Systems: Advances in Post-Pandemic Viral Antigen Production for Vaccines.Int J Mol Sci. 2024 Nov 7;25(22):11979. doi: 10.3390/ijms252211979. Int J Mol Sci. 2024. PMID: 39596049 Free PMC article. Review.
-
Humanized mouse model reveals the immunogenicity of Hepatitis B Virus vaccine candidates produced in CRISPR/Cas9-edited Nicotiana benthamiana.Front Immunol. 2025 Apr 9;16:1479689. doi: 10.3389/fimmu.2025.1479689. eCollection 2025. Front Immunol. 2025. PMID: 40270959 Free PMC article.
-
NAC1 transcriptional activation of LDHA induces hepatitis B virus immune evasion leading to cirrhosis and hepatocellular carcinoma development.Oncogenesis. 2024 May 4;13(1):15. doi: 10.1038/s41389-024-00515-4. Oncogenesis. 2024. PMID: 38704368 Free PMC article.
References
-
- World Health Organization . Global hepatitis report, 2017. (2017), 9789241565455.
-
- World Health Organization . Hepatitis b factsheet (2021). Available at: http://www.who.int/news-room/fact-sheets/detail/hepatitis-b (Accessed 4 May 2022).
-
- World Health Organization . Global health sector strategy on viral hepatitis 2016-2021. towards ending viral hepatitis. Geneva: world health organization; (2016).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

