Proteomics reveals differential adsorption of angiogenic platelet lysate proteins on calcium phosphate bone substitute materials
- PMID: 35936551
- PMCID: PMC9348553
- DOI: 10.1093/rb/rbac044
Proteomics reveals differential adsorption of angiogenic platelet lysate proteins on calcium phosphate bone substitute materials
Abstract
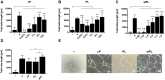
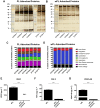
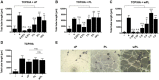
Protein adsorption on biomaterials for bone substitution, such as calcium phosphates (CaP), evokes biological responses and shapes the interactions of biomaterials with the surrounding biological environment. Proteins adsorb when CaP materials are combined with growth factor-rich hemoderivatives prior to implantation to achieve enhanced angiogenesis and stimulate new bone formation. However, the identification of the adsorbed proteins and their angiogenic effect on bone homeostasis remain incompletely investigated. In this study, we analyzed the adsorbed complex protein composition on CaP surfaces when using the hemoderivatives plasma, platelet lysate in plasma (PL), and washed platelet lysate proteins (wPL). We detected highly abundant, non-regenerative proteins and anti-angiogenic proteins adsorbed on CaP surfaces after incubation with PL and wPL by liquid chromatography and mass spectrometry (LC-MS) proteomics. Additionally, we measured a decreased amount of adsorbed pro-angiogenic growth factors. Tube formation assays with human umbilical endothelial cells demonstrated that the CaP surfaces only stimulate an angiogenic response when kept in the hemoderivative medium but not after washing with PBS. Our results highlight the necessity to correlate biomaterial surfaces with complex adsorbed protein compositions to tailor the biomaterial surface toward an enrichment of pro-angiogenic factors.
Keywords: angiogenesis; calcium phosphates; platelet lysate; protein adsorption; proteomics.
© The Author(s) 2022. Published by Oxford University Press.
Figures






Similar articles
-
Characterization of the Protein Corona of Three Chairside Hemoderivatives on Melt Electrowritten Polycaprolactone Scaffolds.Int J Mol Sci. 2023 Mar 24;24(7):6162. doi: 10.3390/ijms24076162. Int J Mol Sci. 2023. PMID: 37047135 Free PMC article.
-
Comparative proteomic analysis of human mesenchymal stromal cell behavior on calcium phosphate ceramics with different osteoinductive potential.Mater Today Bio. 2020 Jun 24;7:100066. doi: 10.1016/j.mtbio.2020.100066. eCollection 2020 Jun. Mater Today Bio. 2020. PMID: 32642640 Free PMC article.
-
Development of proteomic tools to study protein adsorption on a biomaterial, titanium grafted with poly(sodium styrene sulfonate).J Chromatogr B Analyt Technol Biomed Life Sci. 2011 Dec 1;879(31):3681-7. doi: 10.1016/j.jchromb.2011.10.006. Epub 2011 Oct 11. J Chromatogr B Analyt Technol Biomed Life Sci. 2011. PMID: 22036657
-
Systematic approach to characterize the dynamics of protein adsorption on the surface of biomaterials using proteomics.Colloids Surf B Biointerfaces. 2020 Apr;188:110756. doi: 10.1016/j.colsurfb.2019.110756. Epub 2019 Dec 24. Colloids Surf B Biointerfaces. 2020. PMID: 31887650 Review.
-
Novel therapeutic approaches in utilizing platelet lysate in regenerative medicine: Are we ready for clinical use?J Cell Physiol. 2019 Aug;234(10):17172-17186. doi: 10.1002/jcp.28496. Epub 2019 Mar 25. J Cell Physiol. 2019. PMID: 30912141 Review.
Cited by
-
Proteomic Analysis of Human Serum Proteins Adsorbed onto Collagen Barrier Membranes.J Funct Biomater. 2024 Oct 9;15(10):302. doi: 10.3390/jfb15100302. J Funct Biomater. 2024. PMID: 39452600 Free PMC article.
-
Platelet-Rich Plasma in Dermatology: New Insights on the Cellular Mechanism of Skin Repair and Regeneration.Life (Basel). 2023 Dec 25;14(1):40. doi: 10.3390/life14010040. Life (Basel). 2023. PMID: 38255655 Free PMC article. Review.
References
-
- Horbett TA. Protein adsorption on biomaterials. In: Cooper SL, Peppas NA, Hoffman AS et al. (eds). Biomaterials, Interfacial Phenomena and applications. Washington, DC: American Chemical Society, 1982;233–44.
-
- Horbett TA. The role of adsorbed proteins in tissue response to biomaterials. In: Ratner BD (ed). Biomaterials Science: An Introduction to Materials in Medicine. Amsterdam; Boston: Elsevier Academic Press, 2004;237–46.
-
- Bauer TW, Muschler GF.. Bone graft materials. An overview of the basic science. Clin Orthop Relat Res 2000;371:10–27. - PubMed
-
- Kurien T, Pearson RG, Scammell BE.. Bone graft substitutes currently available in orthopaedic practice: the evidence for their use. Bone Joint J 2013;95-B:583–97. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

