Glycyl-L-histidyl-L-lysine-Cu2+ attenuates cigarette smoke-induced pulmonary emphysema and inflammation by reducing oxidative stress pathway
- PMID: 35936787
- PMCID: PMC9354777
- DOI: 10.3389/fmolb.2022.925700
Glycyl-L-histidyl-L-lysine-Cu2+ attenuates cigarette smoke-induced pulmonary emphysema and inflammation by reducing oxidative stress pathway
Abstract
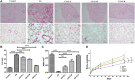
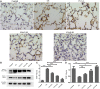
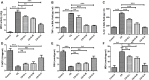
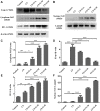
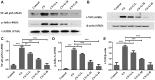
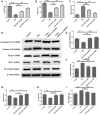
Background: Chronic obstructive pulmonary disease (COPD) is a common respiratory disorder manifested as chronic airway inflammation and persistent airflow limitation with the essential mechanism as inflammatory response and oxidative stress induced by toxic exposures such as cigarette smoke (CS). Glycyl-L-histidyl-L-lysine (GHK) is a nontoxic tripeptide involved in the process of healing and regeneration as a natural product. With the combination of Cu(II), glycyl-L-histidyl-L-lysine-Cu2+ (GHK-Cu) improves antioxidative and anti-inflammatory bioavailability, and they might offer potential therapeutic properties for COPD. Thus, the present study aimed to identify the potential effects of GHK-Cu on emphysema induced by cigarette smoke. Methods: In the in vivo experiment, C57BL/6J mice were exposed to CS for 12 weeks to induce pulmonary emphysema. GHK-Cu was injected intraperitoneally at doses of 0.2, 2 and 20 μg/g/day in 100 µl of saline on alternative days from the 1st day after CS exposure. The effects of GHK-Cu on the morphology of CS-induced emphysema, the inflammatory response and oxidative stress were evaluated. The antioxidative effect of GHK-Cu on human alveolar epithelial A549 cells was assessed in vitro. Results: GHK-Cu treatment attenuated the CS-induced emphysematous changes and partially reversed the matrix metalloprotein -9 (MMP-9)/tissue inhibitor of metalloproteinases-1 (TIMP-1) imbalance in the lung tissue. GHK-Cu reduced the inflammation and oxidation by decreasing the expression of inflammatory cytokines (IL-1β and TNF-α) in the bronchoalveolar lavage and the enzymatic activity of MPO and MDA in the lung homogenate while restoring the T-AOC and GSH content. Furthermore, administration of GHK-Cu reversed the increase in NF-κB expression induced by CS and increased the Nrf2 level, as an antioxidant defense component, in mice with chronic CS exposure. In CSE-exposed human alveolar epithelial A549 cells, GHK-Cu also inhibited oxidative stress by suppressing MDA levels and restoring T-AOC and GSH levels, which were modulated by upregulating Nrf2 expression. Conclusion: GHK-Cu treatment attenuated CS-induced emphysema by anti-inflammation by downregulating NF-κB and antioxidation via upregulation of the Nrf2/Keap1 in lung tissues.
Keywords: GHK-Cu; NF-κB; Nrf2; cigarette smoke; emphysema; inflammation; oxidative stress.
Copyright © 2022 Zhang, Yan, Lu and Zhou.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

